Cisplatin Medication Information
Learn everything you need to know about Cisplatin-pronunciation, uses, dosage guidelines, indications, and when to take or avoid it.
Get up-to-date information on side effects, precautions, warnings, and proper storage to ensure safe usage.
Explore Cisplatin brand names commonly used in India and internationally, along with detailed pricing information. Consult your healthcare provider for tailored medical advice.
Generic Name : Cisplatin Pronunciation : sis' pla tin Therapeutic Classification : ChemotherapyBrand Names or Trade Names of Cisplatin
India :
Cismax 10 mg |
Cisplan (50 mg) |
Cisplatin |
Platin 10MG Injection (1mg/mL) |
Cisteen |
Duplat |
Platin |
Platicis |
Cisteen (50 mg) |
Plationco (50 mg)
International :
Platinol
Why is Cisplatin Prescribed? (Indications)
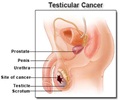
This medication is a platinum-based chemotherapy agent, prescribed for ovarian cancer, bladder cancer and metastatic testicular tumors either alone or with other medications. It stops cancer cell growth.When should Cisplatin not be taken? (Contraindications)
Contraindicated in patients with hearing impairment, kidney impairment and hypersensitivity.What is the dosage of Cisplatin?
Adult: IV- Metastatic ovarian cancer As monotherapy: 100 mg/m2 per cycle, given as a single dose once every 4 week.For combination therapy with cyclophosphamide: 75-100 mg/m2 on day 1 of every 4-week cycle. Metastatic testicular tumors 20 mg/m2/day for 5 days per cycle. Advanced bladder cancer 50-70 mg/m2 per cycle once every 3-4 week.
How should Cisplatin be taken?
It comes as a solution for injection to be administered by a healthcare provider into the vein.What are the warnings and precautions for Cisplatin?
• Caution should be exercised in patients with history of bone marrow suppression, kidney or liver disease, any allergy, who are taking other medications, elderly, children, during pregnancy and breast-feeding.• Monitor complete blood counts, ear and neurological functions regularly while taking this medication.
• Patients may develop with increased risk of nephrotoxicity (kidney damage with irritation), drink adequate fluids to prevent damage.
• Avoid vaccination while taking this medication.
• It may develop new type of cancer; if you have any doubt consult with your doctor regarding this.
What are the side effects of Cisplatin?
Toxic effects - Nephrotoxicity (kidney damage) and ototoxicity (ear damage).Severe effects - Stroke, heart attack, electrolyte disturbances and allergic reactions.
Gastrointestinal - Loss of appetite, nausea and vomiting.
ENT - Ringing in the ear, loss of hearing and deafness.
What are the other precautions for Cisplatin?
Avoid excess dosage.What are the storage conditions for Cisplatin?
This drug is used only under the supervision of physician.Schedule : G
Drugs that are to be used only under medical supervision.
 MEDINDIA
MEDINDIA
 Email
Email