- Messina JP, Humphreys I, Flaxman A, et al. Global Distribution and Prevalence of Hepatitis C Virus Genotypes. Hepatology (Baltimore, Md). 2015;61(1):77-87. doi:10.1002/hep.27259.
- Guidelines for the screening, care and treatment of persons With chronic hepatitis c infection - (http://apps.who.int/iris/bitstream/10665/205035/1/9789241549615_eng.pdf?ua=1)
- Hepatitis C - (http://www.epidemic.org/)
- What is hepatitis C? - (https://www.niddk.nih.gov/health-information/health-topics/liver-disease/hepatitis-c/pages/ez.aspx)
- About Hepatitis C - (https://medlineplus.gov/hepatitisc.html#cat92)
- What to Expect When Getting Tested - Hepatitis C - (http://www.cdc.gov/hepatitis/hcv/pdfs/hepcgettingtested.pdf)
What is Hepatitis C?
Hepatitis C is a chronic contagious viral disease that affects the liver. It is caused by the hepatitis C virus (HCV), that is transmitted through contact with the blood of an affected individual.
Hepatitis viruses include Hepatitis A, B, D and E. The hepatitis A virus (HAV) spreads through contaminated food and water. Infections are mostly mild and can be cured completely. The hepatitis B virus (HBV) spreads through contact with blood and other body fluids. Safe and effective vaccines are present against both HAV and HBV.
In the case of hepatitis C, no vaccine is available against infection. The infection may take years to produce symptoms; most people thus remain unaware of being infected. By the time they become ill and seek help, considerable damage has been done. Early diagnosis is hence vital.
HCV is 10 times more infectious than HIV when comparing direct blood-to-blood transmission.
The rule of ‘twenties’ often describes the fate of patients with hepatitis C:
- About 20% of people infected with hepatitis C virus will completely recover.
- 20% of the rest will develop cirrhosis.
- Of those who develop cirrhosis, about 20% will develop liver cancer.
There are six genotypes (different types) of Hepatitis C virus. Infections due to genotype 1 are common in the United States, South America and Europe, genotype 2 in west Africa, genotype 3 in south Asia including India, genotype 4 in central and north Africa, genotype 5 in South Africa and genotype 6 in SE Asia.
After a person gets infected, around 1012 virus particles get produced each day in the body. The virus can multiply both in the liver as well as the lymphocytes.
Treatment options include anti-viral medications that cause a lot of side effects.
Patients who develop liver cancer or permanent scarring (cirrhosis) of the liver may require a liver transplant.
There is no vaccine at present. Hepatitis C can be avoided with adequate precautions.
The World Health Assembly adopted the first “Global Health Sector Strategy on Viral Hepatitis, 2016-2021” in the month of May 2016. The goal is to eliminate viral hepatitis as a public health problem by the year 2030. The aim is to reduce new infections by 90% and deaths due to the disease by 65%.
Key Facts and Statistics on Hepatitis C
- Hepatitis C is a chronic liver disease caused by the Hepatitis C virus that can cause both the acute and chronic forms of the infection.
- It was originally named a type of non-A non-B hepatitis and was suggested as “Hepatitis C” in the 1970s and proven in 1989.
- World Health Organization (WHO) estimates that 130-150 million people worldwide have the disease.
- 11 million new cases emerged in 2013 alone.
- 1 million cases are detected every year in India.
- Hepatitis C-related liver diseases kill approximately 700,000 people every year.
- In 2013 alone, 343,000 and 358,000 deaths occurred due to liver cancer and cirrhosis respectively.
- It is mostly present in Africa and Central and East Asia.
- It can infect only humans and chimpanzees.
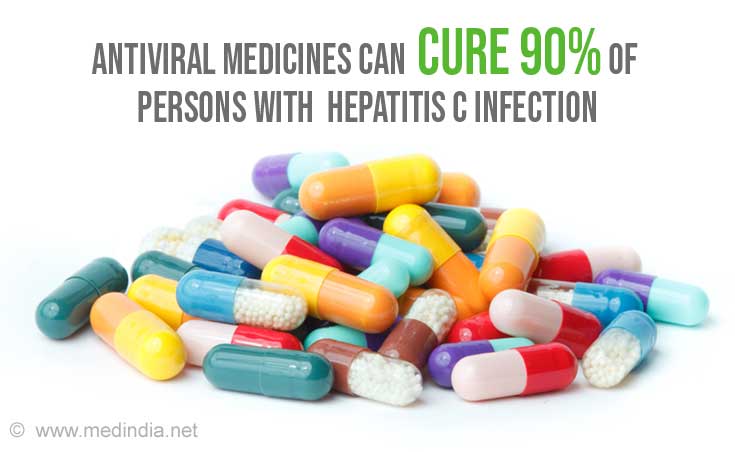
- The infection can be cured with antiviral medicines in 90% of infected persons thereby reducing death due to liver cancer and cirrhosis.

What are the Causes of Hepatitis C infection?
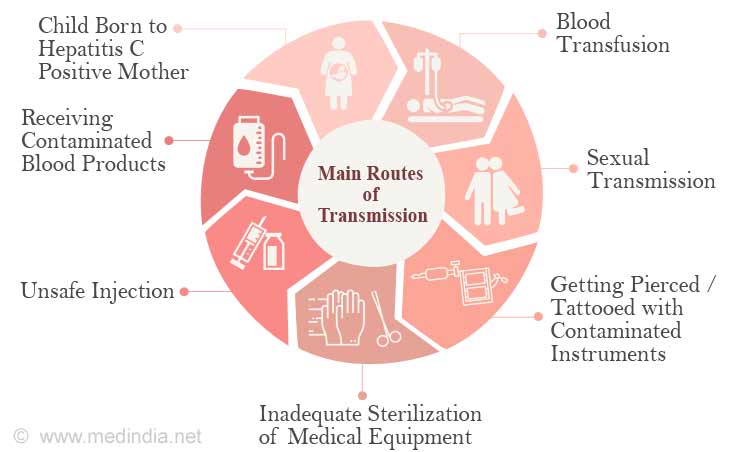
Hepatitis C infection is caused by the hepatitis C virus (HCV).The hepatitis C virus is spread by blood-to-blood contact.
Any practice, activity, or situation that involves blood-to-blood exposure can act as a source of HCV infection.
The following factors increase one’s risk of acquiring HCV:
- Sharing needles, syringes or other equipment (used to inject drugs) with someone who has hepatitis C.
- Receiving blood, blood products, or solid organs from a donor who has hepatitis C.
- Needlestick injuries for workers in healthcare and public settings.
1 out of 20 babies born to mothers with HCV acquires hepatitis C. The risk is higher if the mother has both the Human Immunodeficiency virus (HIV) and HCV.
- In a person who already has HIV infection.
- Spread of HCV by unprotected sexual contact is less common. But people with multiple sex partners, those already having a sexually transmitted disease, or already infected with HIV are at an increased risk for contracting the infection.
- Getting a tattoo or acupuncture in tattoo facilities that follow poor aseptic precautions, and could have contaminated instruments. Licensed commercial tattoo facilities are safer.
- Undergoing long-term kidney dialysis.
- People in prisons where intravenous drug use is common.
- Having elevated liver enzymes (only sign of chronic infection).
- If you were born between 1945 through 1965, chances of having chronic infection is high as blood was not tested for HCV then.
- If you received blood products with clotting factor before 1987.
- Sharing personal items such as toothbrushes and razors with someone who has hepatitis C (the risk is low).
What are the Symptoms and Signs of Hepatitis C infection?
Acute and chronic variants of the disease exist.
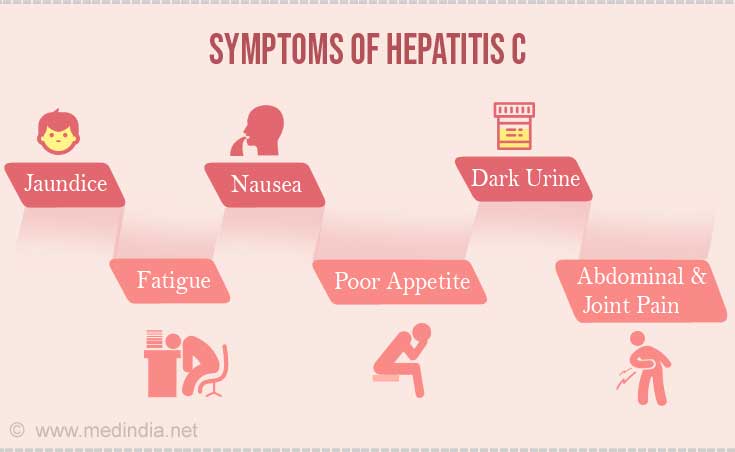
Acute hepatitis C - This is a short-term illness that occurs anytime from 2 weeks to 6 months of getting exposed to the virus. But 80% of people infected with hepatitis C may not show any symptoms at all in the acute phase. Jaundice occurs in about 10% and it gets better.
Some common symptoms that people may exhibit are:
- Abdominal pain especially in the right upper abdomen.
- Abdominal swelling due to collection of fluid; this is called ascites.
- Internal bleeding from the lower part of the esophagus (food pipe). This is due to rupture of dilated veins in the esophagus called varices.
- Dark urine.
- Fatigue - mild or extreme in intensity.
- Fever.
- Itching.
- Jaundice (yellowing of the skin and the whites of the eyes).
- Loss of appetite (anorexia).
- Nausea.
- Pale or clay-colored stools.
- Vomiting.
- Joint pain.
Chronic Hepatitis C - Most people who acquire infection with HCV develop chronic hepatitis. This is a long-term illness and can last a lifetime. The infection may take years or sometimes decades to produce symptoms. It may only be detected due to secondary infections due to liver damage. By the time they become ill and seek help, considerable damage might have been done. Persistence of the infection leads to permanent scarring of the liver (cirrhosis) or liver cancer.
How is Hepatitis C Infection Diagnosed?
1. Screening: Occasionally, hepatitis C gets diagnosed during screening tests, for example during blood donation. Otherwise, routine screening is recommended by WHO for all people who are at high risk.
Abnormal liver enzymes or liver function tests found during routine blood testing may reveal the ailment.
2. Diagnostic Tests:
- Serologic assays like the Enzyme-linked immunoassay (EIA) are performed to detect the presence of antibodies (proteins produced by the immune system to combat antigens) to HCV.
- A non-reactive test result (no HCV antibodies present) means you are not currently infected by the virus.
- A reactive test result means you have been infected with the virus at some point of time. A recombinant immunoblot assay is then performed to confirm this result.
Antibodies to the virus will be present irrespective of the presence of the virus at the point of testing. It is recommended to perform a follow-up test.
- Molecular Assays like the Hepatitis C RNA assays are follow-up tests used to measure virus load or the viral nucleic acid in the body. A number of nucleic acid testing methods like polymerase chain reaction (PCR) test for the presence of virus.
- If there is no RNA (negative result), but the immunoblot is positive, then we can infer that the infection had occurred prior to testing and has been cleared with or without treatment.
- If the immunoblot is negative, it means that the first immunoassay done had wrongly diagnosed the antibodies as it takes about 6-8 weeks after the infection to test positive.
- If there is presence of viral RNA (positive result), then the virus is currently present in the blood.
Viral load tests are also performed during treatment to determine the response to treatment by comparing the viral amount before and during treatment.
3. Genotype assays are usually performed for finding the type of viral genotype that has affected the patient.
4. Liver function tests are carried out to monitor the liver damage. The liver enzyme levels rise at seven weeks after infection but their rise may or may not be an indication of how severe the disease is.
5. Liver biopsy may be ordered if chronic hepatitis C infection is suspected. A small portion of the liver is removed using local anesthesia or a sedative to monitor damage to the liver.
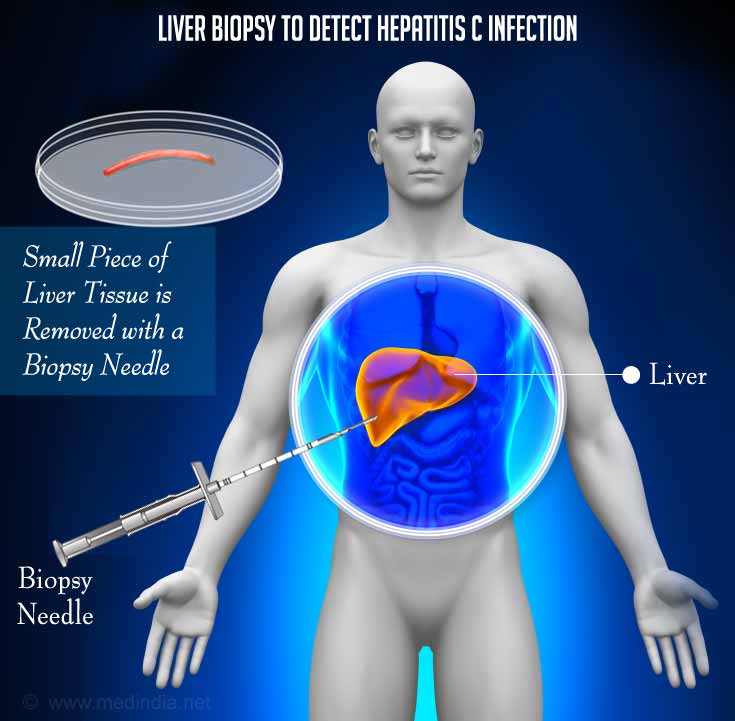
The person will have to lie down with the right arm raised above the head during and about 2 hours after the test. Certain medications might have to be stopped before the procedure and the person might have to fast for 8 hours.
The liver sample is examined under a microscope and analyzed by a doctor for any damage or disease. Changes in bile ducts and lymphocytes are visible.
What is the Treatment for Hepatitis C infection?
40-80% of patients with chronic hepatitis get better with treatment.
Proven hepatitis C virus infection is treated with antiviral medications. Treatment of hepatitis C targets removal of the virus from the blood and should be given to the patients according to the degree of liver scarring.
The duration of treatment is also influenced by the hepatitis C virus genotype. It has been shown that patients with genotypes 2 and 3 are almost three times more likely than patients with genotype 1 to respond to combination therapy.
Prior to 2011, the combination of pegylated interferon-alpha-2a or pegylated interferon-alpha-2b and the antiviral drug ribavirin was usually given for a period of 24 or 48 weeks.
These medications were associated with a number of side effects. Flu-like symptoms occurred in half the patients and a third of them had emotional disturbances.
In 2016, WHO has recommended that direct-acting antiviral (DAA) regimens be used as far as possible instead of regimens with pegylated interferon / ribavirin.
DAA-based regimens have a shorter duration, are given orally, have a lower pill burden (one pill per day), are effective and well tolerated with few adverse effects.
However, pegylated interferon / ribavirin regimens can be used if needed for certain patients. HCV genotype 3 infections with cirrhosis and genotypes 5 and 6 infections with and without cirrhosis are still treated with sofosbuvir / pegylated interferon and ribavirin as an alternative option.
| HCV genotype | Patients without cirrhosis | Patients with cirrhosis |
| 1 | 12 weeks of daclatasvir and sofosbuvirOR12 weeks of ledipasvir and sofosbuvir | 24 weeks of daclatasvir and sofosbuvirOR 12 weeks of daclatasvir / sofosbuvir and ribavirinOR 24 weeks of ledipasvir and sofosbuvir’OR 12 weeks ledipasvir and sofosbuvir and ribavirin |
| 2 | 12 to 16 weeks of sofosbuvir and ribavirin | 16 weeks of sofosbuvir and ribavirin |
| 3 | 24 weeks of sofosbuvir and ribavirin | 24 weeks of daclatasvir / sofosbuvir and ribavirin |
| 4 | 12 weeks of daclatasvir and sofosbuvirOR12 weeks of ledipasvir and sofosbuvir | 24 weeks of daclatasvir and sofosbuvirOR12 weeks of daclatasvir / sofosbuvir and ribavirinOR 24 weeks of ledipasvir and sofosbuvir’OR 12 weeks ledipasvir and sofosbuvir and ribavirin |
| 5 | 12 weeks of ledipasvir and sofosbuvir | 24 weeks of ledipasvir and sofosbuvir’OR 12 weeks ledipasvir and sofosbuvir and ribavirin |
| 6 | 12 weeks of ledipasvir and sofosbuvir | 24 weeks of ledipasvir and sofosbuvir’OR 12 weeks ledipasvir and sofosbuvir and ribavirin |
Liver transplant may be required in cases of liver cirrhosis and liver cancer. The virus recurs in 80-90% of patients who have undergone transplant. This can be decreased by using pegylated interferon and ribavirin post-transplant. The graft could get infected and develop into cirrhosis within 5 years in 10-30% of the people.
Complementary and alternative medicine (CAM) is the use of relaxation and physical (like yoga and tai chi) techniques and consumption of herbal products. Physical and relaxation therapies are generally safe but more research needs to be done to consume herbal products while having liver disease. The National Center for Complementary and Integrative Health (NCCIH) a center of NIH is evaluating various products.
There is no special “hepatitis C diet” but a well-balanced diet can lead to better liver functioning and lowered risk of scarring. It also helps the immune system fight off the illness and keeps the blood sugar under control in case of concurrent diabetes.
What are the Complications of Hepatitis C infection?
Around 70-80% of patients infected with HCV develop chronic infection.
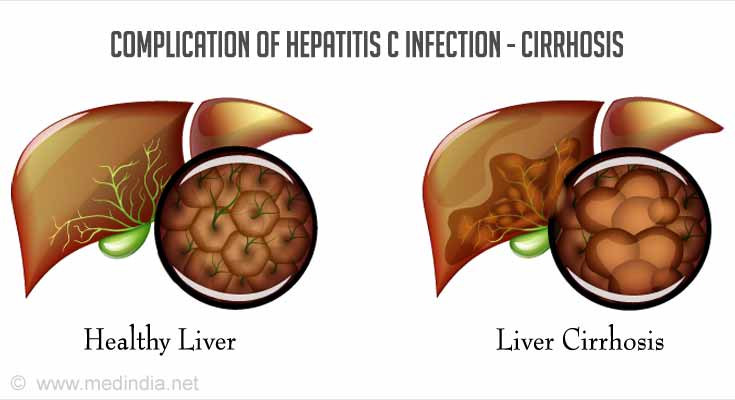
- Permanent scarring of the liver (cirrhosis) occurs within 20 years of disease onset in 20% of those with chronic infection.
- Liver cancer (Hepatocellular carcinoma, HCC) develops in about 1-4% of patients with cirrhosis each year after an average of 30 years.
- Extra hepatic manifestations like cryoglobulinemia (presence of abnormal proteins in blood), membranoproliferative glomerulonephritis (kidney disorders), and idiopathic thrombocytopenic purpura (lowered platelet count without a known cause) can occur.
- Liver failure in the case of advanced cirrhosis.
What is the Prognosis (Outlook) of Hepatitis C infection?
Outlook (prognosis) of hepatitis depends on a number of factors, the most important ones being:
- The amount of virus in the body, i.e. the viral load.
- The genotype of the virus. The advancement of the disease - prognosis gets worse with advancing liver disease.
- Gender - prognosis appears to be better in females with HCV compared to their male counterparts.
- The extent of liver disease.
- Co-existing medical conditions.
- Other influencing factors include like age, ethnicity, general health and response to treatment.
- The majority of patients with chronic hepatitis C will not clear it without adequate management. In a small minority the infection with HCV is self-limited. Cure rates are as high as 60% with the combination therapy.
How Can Hepatitis C Be Prevented?
Hepatitis C is preventable through safe practices. There are no available products that can prevent HCV infection as of now. Vaccines are yet to be developed.
- Avoid sharing needles, razors, toothbrushes, manicure tools or other items that could bear contaminated blood
- Avoid getting pierced or tattooed with non-sterile equipment.
- Use barrier protection (condoms) during sexual intercourse. Practice safe sex.
- Patients with hepatitis C should not donate blood or organs. Exception exists in patients with HCV who require liver transplantation. Studies that support liver transplant recipients getting liver grafts from HCV-positive donors exist.
- Abstain from alcohol use.

 MEDINDIA
MEDINDIA

 Email
Email