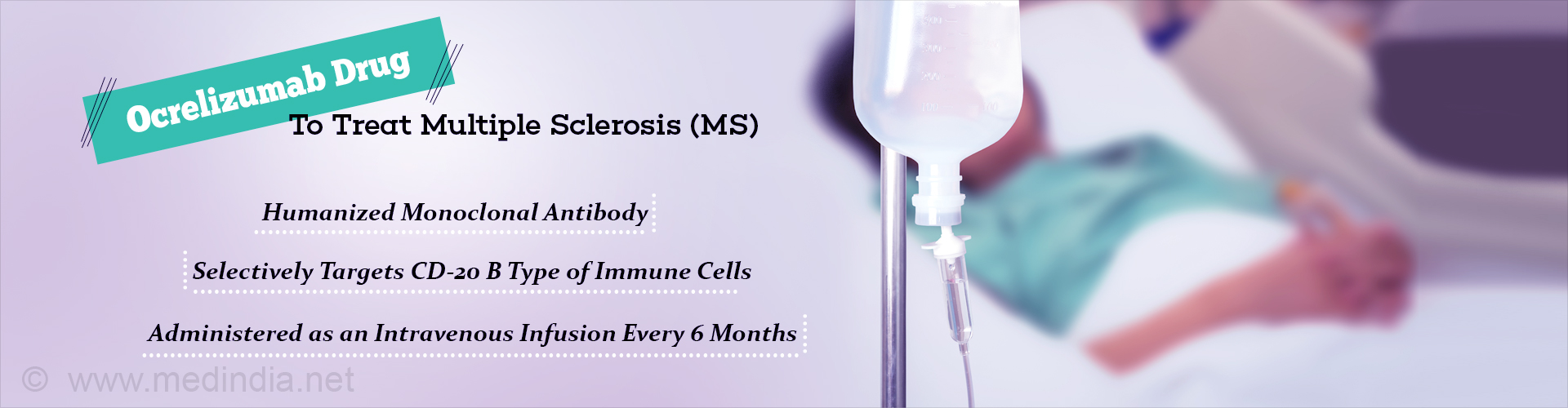
Ocrelizumab drug targets B cells and shows promising results for the treatment of multiple sclerosis.
Highlights
- Multiple sclerosis is a nervous disorder which affects the brain and the spinal cord.
- The efficacy and safety of Ocrelizumab drug were tested to provide treatment options for multiple sclerosis
- Ocrelizumab drug which selectively targets B type of immune cells showed promising results for the treatment of multiple sclerosis.
TOP INSIGHT
New drug Ocrelizumab was found to show positive results in treating multiple sclerosis.
Around 2.3 million people in the world suffer from multiple sclerosis. Symptoms are unpredictable. However, multiple sclerosis may cause fatigue, tremors, problems with balance, problems with memory, concentration and mood changes.
The patients with progressive form of multiple sclerosis who were given ocrelizumab drug showed less medical evidence and brain scan evidence for disease progression when compared to patients who were given placebo drug. The Phase 3 clinical trials were done to test the safety and efficacy of the drug in human patients.
Among patients with relapsing form of multiple sclerosis, ocrelizumab drug showed low rates of disease progression and activity when compared to patients who received interferon beta 1a drug as a standard treatment for multiple sclerosis.
Ocrelizumab drug is under review for the Food and Drug Administration (FDA) approval to treat progressive and relapsing forms of multiple sclerosis.
Dr. Jack Burks, chief medical consultant for the Multiple Sclerosis Association of America, said, "No treatments have been approved by the FDA for primary progressive MS."
Ocrelizumab Drug
Ocrelizumab drug is a human monoclonal antibody that is found to specifically target a type of immune cell called CD20- positive B cells. These cells are key contributors of myelin which is required to protect the nerve cells. The drug is administered in the form of an intravenous infusion for every 6 months.
Peter Chin, group medical director for neuroscience at Genetech, said, "B cells have been targeted in other disease states with other drugs (notably Genentech's Rituxan, which has been used to treat certain blood cancers and rheumatoid arthritis)."
Facts on Multiple Sclerosis
- Multiple sclerosis is a progressive form of a disorder that affects the central nervous system.
- Around 100,000 people in the United Kingdom have multiple sclerosis.
- More number of women are diagnosed with multiple sclerosis when compared to men.
- People between the age of 20 and 30 are usually diagnosed with multiple sclerosis.
- Multiple sclerosis is not a genetic disorder, but patients are at a higher risk of having multiple sclerosis.
- Facts and myths about MS - (https://www.mstrust.org.uk/understanding-ms/what-ms/facts-and-myths-about-ms)
- Ocrelizumab - (https://www.mssociety.org.uk/ms-research/treatments-in-the-pipeline/ocrelizumab)
- Multiple Sclerosis - ( https://medlineplus.gov/multiplesclerosis.html )
- Multiple Sclerosis: Just the Facts - (https://www.nationalmssociety.org/NationalMSSociety/media/MSNationalFiles/Brochures/Brochure-Just-the-Facts.pdf)
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email










