A promising new test to identify patients with a rare liver disease who will not respond to standard treatment has been developed by researchers.
TOP INSIGHT
The newly developed NanoString nCounter digital expression platform provides a reliable indicator of a primary biliary cholangitis patient's likelihood of failing to respond to the standard treatment.
The new test has identified a gene signature related to the immune response in the liver tissue of patients with high risk disease, including the senescence marker p21WAF1/Cip.
Using the NanoString nCounter digital expression platform, standard liver biopsies taken at diagnosis can be directly and rapidly screened for this gene signature.
The team are looking to work with industry to develop this as a companion diagnostic test which alongside a new treatment would determine its applicability to a specific patient.
Publishing the work in e-biomedicine, lead author Dr Lucy Walker from the Institute of Cellular Medicine at Newcastle University said, "This NanoString test would be easy to introduce into a clinical setting as it needs no separate samples. Our initial results show the technique provides a reliable indicator of a patient's likelihood of failing to respond to the standard treatment. This means medical teams could intervene earlier with alternative treatments, increasing the chances of success and perhaps staving off the need for a liver transplant."
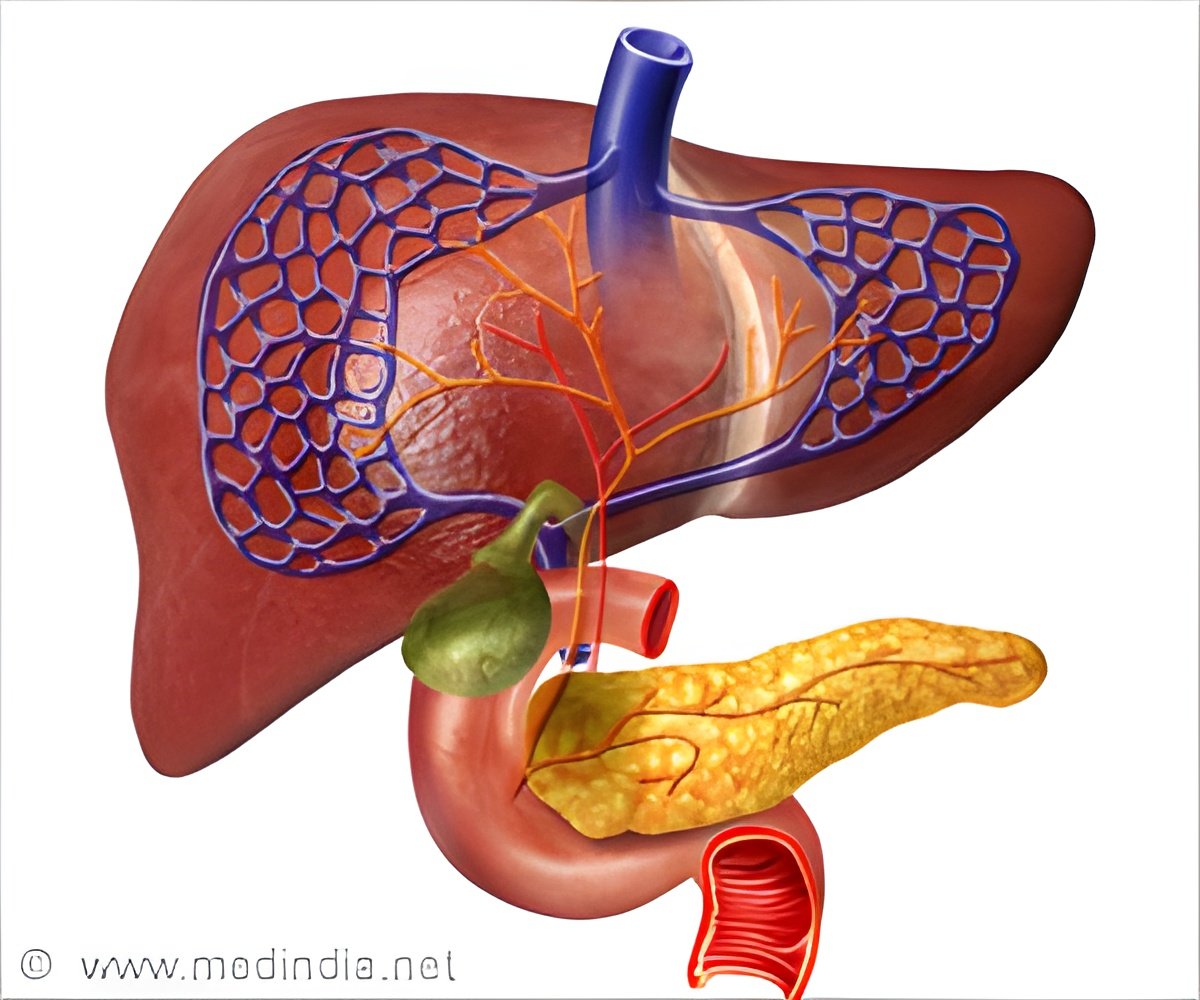
In patients with PBC, the body thinks that the bile ducts within the liver are foreign objects and tries to destroy the lining to these ducts. These bile ducts are designed to allow the flow of bile from the liver, so damage of these ducts leads to poor drainage of bile acids.
PBC is relatively rare affecting up to one in 3-4,000 people and of those nine out of 10 are women.
While there is a genetic element to PBC, it is thought to be within the auto-immune family of conditions and experts do not yet know what causes PBC or what can cure it.
In the early stages of the disease, PBC often doesn't cause any symptoms and many people are only diagnosed during tests carried out for another reason.
Testing
Developed at the NanoString facility within the Faculty of Medical Sciences at Newcastle University, the test raises questions about PBC as lead author, Dr Lucy Walker, explains: "As we have identified a separate gene expression for those with high-risk PBC, it raises the possibility that PBC may actually be two separate conditions with very similar symptoms. This is something we will be examining further in future, larger studies."
This study only examined samples from a small group of patients (13) and a further study into a larger group to validate the results will begin shortly. The work was funded by the Medical Research Council.
Dave Jones, Professor of Liver Immunology and co-author on the paper said: "Currently patients with high-risk PBC may be offered immunotherapies, however, these have mixed results and it may be that often the aggressive nature of the condition in high-risk patients means that even as the treatment starts the condition has created too much damage in the liver. A test which could identify those who would benefit from treatment and provide earlier intervention may increase the possibilities of success."
This test has emerged from the unique collaboration of healthcare expertise and research excellence at the CRESTA clinics (Clinics for Research and Service in Themed Assessment) which are led and managed by the Newcastle Hospitals and bring in expertise from Newcastle University and are supported by the NIHR Newcastle Biomedical Research Centre.
The CRESTA clinic provides enhanced care for patients with PBC through a 'one-stop' multidisciplinary visit to better serve the needs of a diverse patient population, ranging from young working mums to older patients with complex needs. The clinic is also geared for recruitment of such patients for participation in early phase trials - Newcastle is one of the world's leading centers for trials of new therapy in PBC.
This is part of the Newcastle Academic Health Partners, a collaboration involving Newcastle Upon Tyne Hospitals NHS Foundation Trust, Northumberland, Tyne and Wear NHS Foundation Trust and Newcastle University. This partnership harnesses world-class expertise to ensure patients benefit sooner from new treatments, diagnostics and prevention strategies.
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email