Immunoscore an effective tool that predicts which patients are at high risk of tumor recurrence among colon cancer patients has been developed by experts. Immunoscore will help improve the care and treatment of colon cancer.
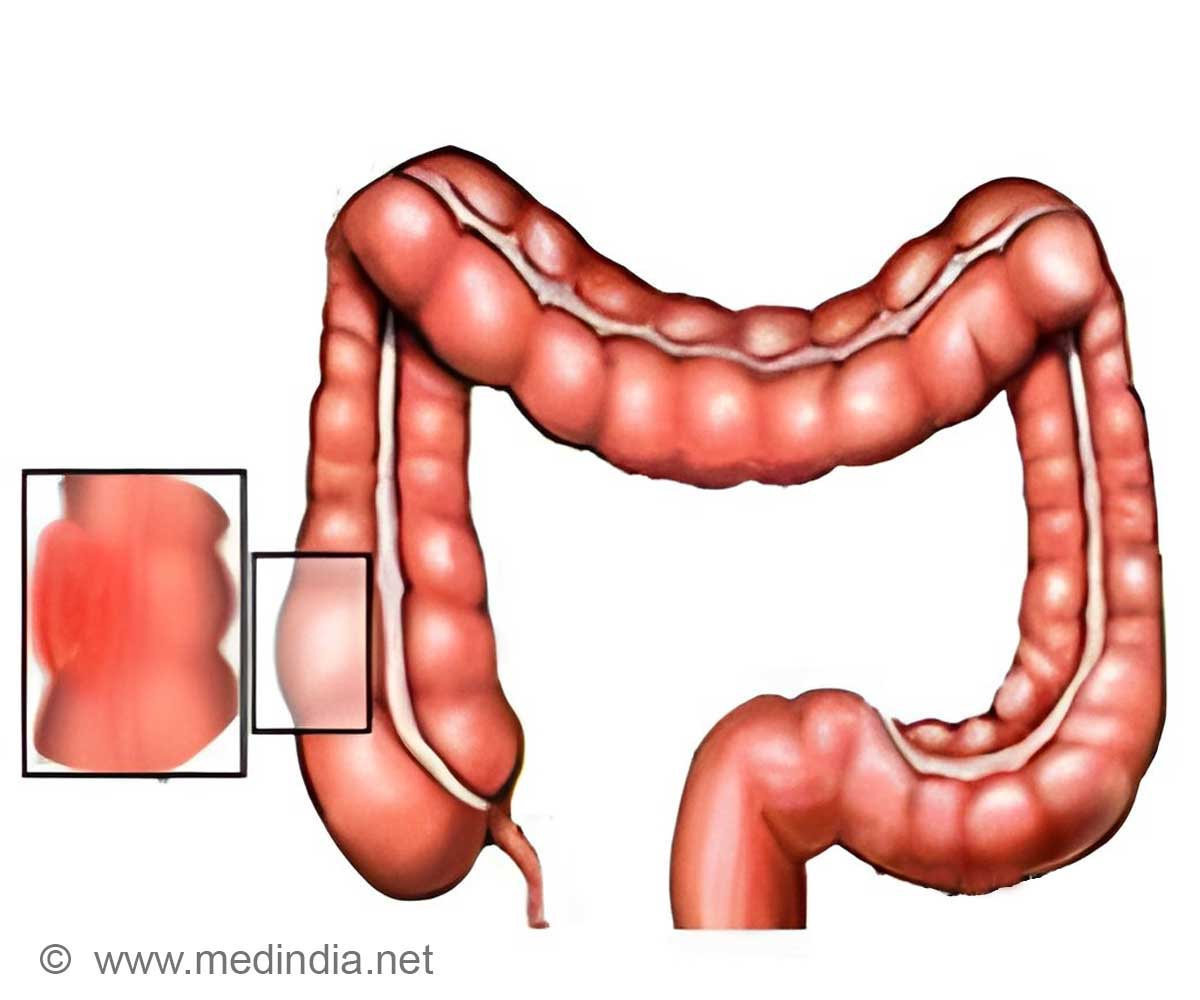
- A new detection tool for colon cancer has been developed.
- It is called the ‘immunoscore’.
- It can identify patients at risk of tumor recurrence.
TOP INSIGHT
A new test tool called ‘immunoscore’ detects disease progression in patients with colon cancer, identifying which patient is at risk of tumor recurrence.
For decades, it has been thought that the immune reaction developed by the patient against his or her cancer has a beneficial influence. Researchers from Inserm and doctors from the Paris AP-HP hospitals have demonstrated in recent years that infiltration of the cancerous tumor by immune cells is a good indication of the way in which colorectal cancer might develop, thereby representing a potential prognostic tool. The immune cell populations which provide the most information on disease progression were identified and the method of evaluating these populations optimized.
This method has led to the creation of an immunological test, applicable in clinical practice, called "Immunoscore". It works by quantifying the density of two types of immune cells in the tumor and its invasive margin: total T-cells (CD3+) and killer T-cells (cytotoxic CD8+).
The objective of this international study published in The Lancet was to evaluate the prognostic value of Immunoscore in patients with colon cancer on a very large scale. For this, an international consortium of 14 immunology and pathology centers in 13 countries was formed. A total of 2,681 patients from these centers were included in this analysis. The prognostic performance of Immunoscore, in which patients are classified into 3 groups (high, intermediate and low), was evaluated on the basis of recurrence risk (evaluated during the 5 years following the surgery) and survival. The statistical analyses were all performed by a group of external biostatisticians from the Mayo Clinic in the USA. The results show that patients with a high Immunoscore present the lowest recurrence risk and prolonged survival.
In the test group comprising 700 patients, only 8% of those with a high Immunoscore presented a recurrence after 5 years. However, the recurrence rate increased significantly in patients with intermediate and low Immunoscores, reaching 19% and 32%, respectively. These findings were confirmed in the two other patient groups analyzed, representing 1,981 patients. Furthermore, Immunoscore had a stronger bearing on patient survival than the tumor criteria which are currently used to guide therapy.
In view of the highly positive results of this test in colon cancer, researchers are currently evaluating Immunoscore in other types of cancer and are studying its ability to predict patient response to the immunotherapies which are currently revolutionizing the treatment of cancer.
- International Validation of The Consensus Immunoscore for The Classification of Colon Cancer: A Prognostic and Accuracy Study - ( https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(18)30789-X/fulltext)
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email