Ivosidenib Medication Information
Discover comprehensive details about Ivosidenib, including its pronunciation, uses, dosage instructions, indications, and guidelines on how and when to take it or avoid it.
The updated prescription information covers potential side effects, precautions, warnings, and storage recommendations.
Additionally, explore the Ivosidenib brands available in India and internationally, along with pricing information. For personalized advice, consult your healthcare provider.
Generic Name : Ivosidenib Pronunciation : EYE-voe-SID-e-nib Therapeutic Classification : ChemotherapyBrand Names or Trade Names of Ivosidenib
India :
Tibsovo
Why is Ivosidenib Prescribed? (Indications)
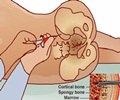
Ivosidenib tablets for oral use are prescribed to treat acute myeloid leukemia (AML). AML is a cancer of the bone marrow and blood in adult patients. Ivosidenib is used in patients who have shown resistance to other cancer medications or whose cancer gets worse after treatment.Ivosidenib treats AML caused by a mutation or a permanent gene alteration in IDH1 which is confirmed by Real Time IDH1 Assay, an FDA-approved test to detect IDH1 mutations.
When should Ivosidenib not be taken? (Contraindications)
Ivosidenib should not be used in patients-• Allergic to ivosidenib
• Pregnancy and breastfeeding
• Guillain-Barré syndrome (Nerve disorder)
• Children under 18 years of age because of the lack of safety and effectiveness data in these age groups
• Undergoing treatment to correct infertility
Caution is required in treating patients with-
• Irregular heartbeat
• Elderly patients
• History of muscle weakness
What is the dosage of Ivosidenib?
• The recommended dose of ivosidenib tablets is 500 mg taken once daily and continued until the patient shows unacceptable toxicity or progression of the disease.• Ivosidenib can be continued for a minimum of 6 months in patients who never show disease progression or when the treatment with drug does not result in intolerable toxicity.
• The dose of ivosidenib can be temporarily stopped or reduced to 250 mg once daily, if abnormal heart rhythm, nerve damage, and symptoms of differentiation syndrome such as an increase in the number of white blood cells occur.
• Treatment with the usual adult dose can be started once the side effects are mild or reduced greatly.
• Similarly, treatment with ivosidenib can be permanently stopped if the symptoms and signs are life-threatening.
How should Ivosidenib be taken?
• Ivosidenib comes as a tablet that can be taken by mouth either with or without food.• Ivosidenib tablets should be taken at the same time each day as prescribed by the doctor.
• Swallow the ivosidenib tablet as a whole with a sufficient quantity of water. Do not crush or split the tablet contents.
• Avoid taking ivosidenib tablets with a high-fat meal as it may increase the availability of ivosidenib which may result in a serious abnormal heart rhythm.
• Never take an extra or another dose if vomiting occurs after taking an ivosidenib tablet but continue with the next scheduled dose.
• If a dose of ivosidenib is missed, administer the missed dose as soon as possible within 12 hours from the missed dose.
• If the next dose is within 12 hours, skip the missed dose and continue with the next dose.
• Taking two doses of ivosidenib tablets within 12 hours is not advised.
What are the warnings and precautions for Ivosidenib?
• Ivosidenib can cause irregular heartbeat and patients must be regularly monitored by assessing the electrolytes level and ECG. Discontinue ivosidenib treatment if any symptoms and signs of abnormal heart rhythm occur.• Patients may experience symptoms of differentiation syndrome such as fever, difficulty in breathing, low blood pressure, lung infection, and rashes along with high levels of white blood cells or leukocytosis anywhere between the first day and three months of ivosidenib treatment.
• Differentiation syndrome can be managed by administering dexamethasone 10 mg directly into the vein every 12 hours.
• If the presence of non-infectious leukocytosis is confirmed, patients should be treated with hydroxyurea or leukapheresis, a laboratory procedure to remove white blood cells from the blood.
• The dose of steroids (dexamethasone) and hydroxyurea can be reduced gradually for three days when the symptoms and signs of differentiation syndrome with leukocytosis are under control.
• If the side effects do not subside within 48 hours even after initiating steroid treatment, stop ivosidenib treatment temporarily until the side effects are reduced.
• Monitor the patients for any new signs or symptoms such as muscle weakness, burning or prickling sensation, numbness and pain either one side or both side of the body, changes in taste, vision or hearing during treatment with ivosidenib.
• During such situations treatment with ivosidenib should be permanently stopped; otherwise patients are vulnerable to develop a rare neurological autoimmune disorder called Guillain Barré syndrome.
What are the side effects of Ivosidenib?
Gastrointestinal: Stomach pain, diarrhea, nausea, vomiting, constipation, mucositis, loss of appetiteNervous system: Fever, headache, fatigue, numbness and pricking sensation, changes in taste, vision, touch or hearing
Cardiovascular: Irregular heart rhythm, reduction in blood pressure, fainting while standing suddenly, fluid accumulation, swelling of legs
Respiratory: Chest pain, cough, breathing difficulty, fluid buildup around lung cavity
Musculoskeletal: Muscle pain, muscle weakness, pain in the joints, neck pain, muscle stiffness, pain in the legs or hands
Others: Rashes, abnormal lab values of hemoglobin, liver enzymes, sodium, magnesium, potassium, uric acid, phosphate, and bilirubin
What are the other precautions for Ivosidenib?
• A blood test should be performed before and during the treatment with ivosidenib to detect any blood disorders. In such cases, the dose can either be reduced or the treatment can be stopped temporarily or permanently.• Ivosidenib can affect fertility in men and women of reproductive potential; therefore patients must be advised about sperm or egg banking preservation for later use when they decide to have a child.
What are the Drug Interactions of Ivosidenib?
• Antifungal drugs such as itraconazole or ketoconazole should not be taken with ivosidenib as the latter results in loss of antifungal activity.• Ivosidenib may decrease the effects of hormonal contraceptives and patients must be advised to rely on additional or alternative contraceptive methods to avoid unwanted pregnancy.
• Ivosidenib can cause abnormal heart rhythm; therefore avoid taking ivosidenib with drugs that cause an irregular heartbeat to prevent life-threatening arrhythmia.
• Rifampicin or rifampin should not be taken along with ivosidenib because the effect of ivosidenib is reduced which may result in treatment failure.
What are the storage conditions for Ivosidenib?
• Store ivosidenib tablets at room temperature between 20°C and 25°C.• Protect from excess moisture and direct sunlight.
• Keep the ivosidenib tablets away from the reach of children and pets.
 MEDINDIA
MEDINDIA
 Email
Email