Baricitinib Medication Information
Discover comprehensive details about Baricitinib, including its pronunciation, uses, dosage instructions, indications, and guidelines on how and when to take it or avoid it.
The updated prescription information covers potential side effects, precautions, warnings, and storage recommendations.
Additionally, explore the Baricitinib brands available in India and internationally, along with pricing information. For personalized advice, consult your healthcare provider.
Generic Name : Baricitinib Pronunciation : bar-i-SYE-ti-nib Therapeutic Classification : ImmunosuppressantsBrand Names or Trade Names of Baricitinib
India :
Olumiant
Overview of Baricitinib
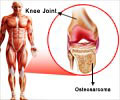
Baricitinib tablets for oral use were approved by FDA on May 31, 2018, to use in patients with rheumatoid arthritis who failed to show a response with other drugs.Why is Baricitinib Prescribed? (Indications)
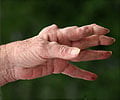
Baricitinib tablets are prescribed to treat adult patients suffering from moderate to severe rheumatoid arthritis who show loss of response to one or more tumor necrosis factor antagonist treatment schedules.Baricitinib works by blocking the actions of Janus kinase (JAK) which is involved with the activation of signaling of molecules and the proinflammatory mediators which are found to be responsible for causing autoimmune disorders.
Baricitinib is recommended for use either alone or in combination with methotrexate or with other disease-modifying antirheumatic drugs (DMARDs).
The European medicines agency (EMA) approved baricitinib tablets to use throughout the EU on Feb 13, 2017.
When should Baricitinib not be taken? (Contraindications)
Baricitinib should not be used in patients with-• Allergy to baricitinib
• Pregnancy and breastfeeding
• History of active or severe infection (e.g., Tuberculosis)
• Blood disorders (e.g., Anemia, low lymphocyte or neutrophil count)
• Severe kidney or liver disease
Baricitinib tablets should not be used in children below 18 years of age as their safety and effectiveness have not been studied in the group.
Caution is required while treating patients with-
• History of malignancy or cancer
• Peptic ulcer or diverticulitis
• Presence of blood clots (Deep vein thrombosis, Pulmonary embolism)
What is the dosage of Baricitinib?
• The recommended adult dose of baricitinib is 2 mg once a day.• The maximum dose of baricitinib tablets is 4 mg and can be reduced to 2mg if the disease comes under control.
• Dose adjustments or modifications should be considered in patients with serious infections or blood disorders such as a decrease in the count of hemoglobin, lymphocytes, and neutrophils.
• Treatment with baricitinib should be withheld in case of any occurrence of severe infections and must not be continued until the infection is brought under control.
How should Baricitinib be taken?
• Barcitinib is available in tablet form which should be taken by mouth either with or without food.• Take the tablet at any time of the day but stick to the same time each day.
• Swallow the tablets as a whole with sufficient quantity of water.
What are the warnings and precautions for Baricitinib?
• Avoid taking live vaccines during baricitinib treatment as it may affect the effectiveness of the vaccines.• Patients should be advised to check their lipid levels after 12 weeks of baricitinib treatment as there have been reports of abnormal levels of liver enzymes and lipid parameters such as total cholesterol and low-density lipoproteins (LDL) during baricitinib treatment.
• Decreased levels of neutrophils, hemoglobin, and lymphocytes can occur during baricitinib treatment, and dose modifications or adjustments are recommended in such cases. In case of abnormally low levels, baricitinib treatment should be immediately stopped and the patient should be treated for blood disorders.
• Stomach pain or related symptoms that occur during baricitinib treatment should be evaluated immediately to detect the presence of any gastrointestinal perforation and to get treated early.
• Patients with a strong history or at an increased risk of acquiring skin cancer should perform periodic skin examination during baricitinib treatment to detect and treat cancers such as non-melanoma skin cancer(NMSC).
• Screening for viral hepatitis should be done before starting treatment with baricitinib as the drug may cause viral reactivation.
• Treatment should be stopped immediately if a patient develops herpes zoster during the treatment with baricitinib.
• Patients should be examined for the presence of active or latent tuberculosis (TB) and should be treated first with antimycobacterial therapy before initiating baricitinib therapy if the patient tests positive for TB.
• Patients should be monitored for the presence of any symptoms or signs of tuberculosis during baricitinib therapy as the infection can develop even in patients who tested negative for TB before starting the therapy.
What are the side effects of Baricitinib?
Gastrointestinal: Nausea, stomach pain, gastritis, ulcer, gastroenteritis or infectious diarrhea, high levels of blood cholesterol, low-density lipoproteins (LDL), and abnormal liver enzymes (AST (SGOT) and ALT (SGPT)) levelsRespiratory system: A sore throat, obstruction of the nose, sinusitis, common cold, swelling of the tonsils and pharynx
Blood: Low levels of hemoglobin, neutrophils, and lymphocytes, blood clots
Kidney: Urinary tract infections such as cystitis and pyelonephritis
Others: Viral infections such as herpes simplex or herpes zoster
What are the other precautions for Baricitinib?
• Patients or caregivers should consult the physician immediately if any signs or symptoms of infection occur during baricitinib therapy who can suspend the treatment until the infection is controlled.• Never use baricitinib tablets for reasons it is not prescribed for and do not recommend it to another patient who reports with the same symptoms as you.
What are the Drug Interactions of Baricitinib?
• Immunosuppressant drugs (e.g., Azathioprine, Cyclosporine, Tacrolimus) or JAK inhibitors (e.g., Tofacitinib, Ruxolitinib) should be avoided in combination with baricitinib tablets as it may result in additive immune suppression.• Non-steroidal antiinflammatory drugs such as diclofenac or ibuprofen should not be used together with baricitinib as they may increase the concentration of baricitinib or pose a risk of gastric perforation and must be avoided.
Other medications that should not be used together or require caution when used with baricitinib are as follows:
• Probenecid
• Corticosteroids
• Leflunomide or teriflunomide
What are the storage conditions for Baricitinib?
• Baricitinib tablets should be stored at a controlled room temperature of 20°C to 25°C.• Protect the tablets from excess heat and moisture.
• Keep baricitinib tablets out of reach of children.
 MEDINDIA
MEDINDIA
 Email
Email