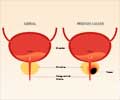
To monitor the effects of treatment, researchers tracked variables like Progression Free Survival, and changes in blood levels of prostate-specific antigen.
To monitor the effects of treatment, MD Anderson researchers tracked several variables including Progression Free Survival, as well as changes in blood levels of prostate-specific antigen (PSA) and bone-specific alkaline phosphatase (BAP, a marker of prostate cancer in bone cells). In addition, safety and toxicity were monitored for both patient groups.
Analysis and comparison of the data demonstrated that median PFS was significantly longer for patients receiving combination versus single agent chemotherapy (6.7 months vs 4.4 months, respectively, p = 0.01). Furthermore, reductions in both PSA and BAP were greater for the combination therapy group. PSA reductions greater than 50 percent occurred 60 percent of the time with combined chemotherapy vs. 44 percent with the single drug. PSA reductions greater than 90 percent occurred 28 percent of the time with two chemotherapy drugs vs. 20 percent with one. In addition, BAP reductions greater than 50 percent for combination vs. single drug were 63 percent and 25 percent respectively.
Side effects, such as fatigue, anemia and neutropenia were comparable for both the single-drug regimen and two-drug regimen. In addition, there were no significant toxicity events.
"We believe cabazitaxel-carboplatin combination chemotherapy may become the clinical standard for advanced prostate cancer once additional safety, efficacy and overall survival data is generated," explained Paul Corn, M.D., Ph.D., an associate professor of genitourinary medical oncology at MD Anderson. "Dr. Ana Aparicio’s lab is currently developing tumor-specific biomarkers to identity patients with an aggressive variant of prostate cancer most likely to benefit from this approach."
Source-Eurekalert