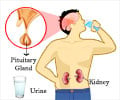
The mitochondrial destruction related oxidative stress within the endothelial cells of the glomerulus, progressively impairing kidney function.
- Diabetic kidney disease is the major cause of death in diabetic patients due to kidney failure.
- The mitochondria within the endothelial cells of the glomerulus of the affected kidney produces excess amounts of reactive oxygen species (ROS).
- These molecules damage the proteins, DNA and other cell types in the glomerulus, eventually leading to its destruction and kidney failure.
An often fatal complication of diabetes is kidney disease. It is difficult to diagnose in early and if diagnosed early it is treatable.
According to the National Kidney Foundation, around 30% of patients with type 1 (juvenile onset) diabetes and 10% to 40% of those with type 2 (adult onset) diabetes eventually will suffer from kidney failure. Patients must then turn to dialysis or kidney transplantation.
In United States, kidney disease is the 9th leading cause of death in the United States and an estimated 31 million people have chronic kidney disease (CKD).
Diabetes is the leading cause of kidney failure. Diabetes causes 44% of all new cases of kidney failure.
"Diabetic kidney disease is one of the major causes of death in diabetic patients, and is also the leading single cause of end-stage renal disease in the United States," says the study's senior investigator, Ilse S. Daehn, PhD, Assistant Professor of Medicine (Nephrology) at the Icahn School of Medicine at Mount Sinai. "Our findings open new diagnostic opportunities for early detection and potential therapeutic strategies to protect against further renal damage in patients."
Destruction of Glomerulus
Researchers focused on the kidney's glomerulus which are key unit in the filtration of blood for waste products to be expelled in urine.
They used two groups of mice to study three different cell types that interact within the glomerulus.
One group naturally develops diabetic kidney disease and the other group is naturally resistant to the disorder.
In mice prone to kidney disease, the wafer-like endothelial cells that formed the inner lining of blood vessels were affected.
The mitochondria which are the cellular subunits that act like power plants, were stressed, and so made excess amounts of reactive oxygen species (ROS). When over-produced, these molecules can damage cell proteins and DNA.
This process begins to destroy podocytes, cells that wrap around and work with capillaries and the other cell types in the glomerulus.
The glomerulus eventually becomes brittle, the capillaries collapse, and kidneys become leaky, shedding essential body proteins.
Progressive damage leads to kidney failure, resulting in end-stage kidney disease.
Cellular Receptor
Researchers discovered a pathway within the endothelium in the glomerulus. This pathway helped to manage oxidative stress triggered by reactive oxygen species (ROS).
This pathway produced excess quantities of a cell receptor, endothelium receptor-A, as well as its ligand. Ligand is the protein that binds to the receptor.
Researchers then inferred that developing a small molecule that blocks the ligand from binding to its receptor, might reduce production of mitochondrial ROS. This will halt the resulting damage to the glomerulus.
On using an experimental small molecule, BQ-123, to specifically block this receptor in mice who would naturally develop the diabetic kidney disease, they found that this molecule stopped the disease progression.
Researchers confirmed their hypothesis by testing urine and kidney biopsies from patients with diabetic kidney disease.
They found molecules in the urine linked to oxidative stress and rapid disease progression, and biopsies that showed increased mitochondrial DNA damage and increased endothelium receptor-A expression.
"These findings in human samples go a long way to substantiate our hypotheses, which is exciting because it represents a new way forward to understanding and treating diabetic kidney disease," Dr. Daehn says.
The study is published in Diabetes.
References
- 2015 KIDNEY DISEASE STATISTICS - (http://www.kidneyfund.org/assets/pdf/kidney-disease-statistics.pdf)
- Ilse S. Daehn et al. Glomerular Endothelial Mitochondrial Dysfunction Is Essential and Characteristic of Diabetic Kidney Disease Susceptibility . Diabetes; (2017) doi.org/10.2337/db16-0695
Source-Medindia