White blood cell boosting drugs had no negative impact during concurrent chemo-radiotherapy for lung cancer, reveals study.
Highlights
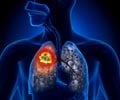
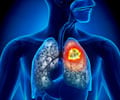
- Lung cancer occurs due to the uncontrolled growth of cancer cells in the lungs.
- The optimal treatment for lung cancer is concurrent chemoradiotherapy, but its efficacy is impeded by increased toxicity in blood, esophagus and the lungs.
- A recent trial analysis presented at the European Society for Medical Oncology found white blood boosting drugs to be safe during chemoradiotherapy.
Granulocyte colony-stimulating factors (G-CSFs) are used as a supportive measure for boosting the survival, proliferation and differentiation of neutrophils.
Neutropenia (Decreased count of the neutrophils) could be less severe and patients may recover more quickly and reduce the risk of infectious complications.
However, due to the use of concurrent chemoradiotherapy in small cell lung cancer is controversial. The American Society of Clinical Oncology recommends against its routine use.
Clinical Trial
The study has been conducted in a randomized trial with 215 patients between 1989 and 1991. The trial results showed that there was a significant increase in severe thrombocytopenia, severe anemia, pulmonary complications and toxic deaths when the Granulocyte-macrophage CSFs (GM- CSFs) were used during chemotherapy.
In the phase III CONVERT Trial, around 547 patients were enrolled for limited-stage lung cancer chemoradiotherapy. These patients had no difference in the overall survival between the two groups.
Trial Findings
The findings confirmed that the chances of severe thrombocytopenia or anemia had been doubled nearly 30% and 20% respectively in patients who were given G-CSF.
These rates were lower than that which was previously reported. The levels were measured following a significantly higher use of supportive measures such as platelets and blood transfusions.
The study results also showed that there was no difference in the incidence of pulmonary complications or survival.
Gomes said, "G-CSF had no significant negative impact on the outcomes of these patients, which is a very comforting result. The higher hematological toxicity was balanced by an appropriate supportive care throughout treatment."
"We can conclude from this analysis that the use of G-CSF during thoracic radiotherapy is safe and should support patients to receive the full planned course of concurrent chemoradiotherapy and achieve the best possible benefit. These findings should give clinicians the confidence to use G-CSF when needed in this context. We aim to publish a complete analysis later this year which may hopefully help change the current guidelines," Gomes said.
Dr Stefan Zimmermann, Senior Consultant, Medical Oncology Department, HFR - Hôpital Cantonal, Fribourg, Switzerland commented that, "Oncologists do need G-CSF to mitigate neutropenia and increase chemotherapy delivery and compliance, but want the beneficial effect of timely concurrent therapy to outweigh the toxic risks."
"In this analysis, the use of G-CSF did not result in an increased risk of pneumonitis, but the incidence of severe thrombocytopenia is a concern," he continued.
"The use of G-CSF was not detrimental to progression-free survival or overall survival. We can conclude that primary or secondary prophylaxis of febrile neutropenia with G-CSF is justified, but patients at higher risk for thrombocytopenia should be treated with caution."
Source-Medindia