Vitamin D supplementation at high doses for over 5 years does not provide any additional protection against cancer or cardiovascular disease (CVD), reveals a new study.
- The study investigates whether vitamin D supplementation reduces the risk of cancer or cardiovascular disease (CVD)
- Even after vitamin D supplementation at high doses for over 5 years, the incidence of cancer or major cardiovascular events was not reduced
- Prevention of the incidence of cancer or CVD by supplementation with high doses of vitamin D is not possible in a diverse, multi-ethnic US population
TOP INSIGHT
Vitamin D supplementation at high doses for over 5 years does not decrease the incidence of cancer or CVD, compared to a placebo. The study cohort consisted of a diverse population of nearly 26,000 participants from USA. Prevention of cancer or CVD by vitamin D supplementation is not possible in the US population.
Read More..
Study Background
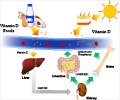
In recent years, it has been suggested that vitamin D supplementation plays a role in the prevention of cancer and CVD. However, it is not certain whether supplementation of vitamin D prevents cancer or CVD, since confounding factors like outdoor activities (increased sun exposure), adiposity (decreased bioavailability of 25-hydroxyvitamin D), and nutritional status, amongst others, may produce false protective associations.Study Objective
The objective of the study was to investigate whether vitamin D supplementation reduces the risk of cancer or CVD, as very little data is available from randomized clinical trials. This trial is called the Vitamin D and Omega-3 Trial (VITAL), which is a large-scale trial for evaluation of high-dose vitamin D and omega-3 fatty acids.Study Design
This was a randomized, placebo-controlled clinical trial with a two-by-two factorial design that was conducted across the US. The trial evaluated the role of vitamin D3 (cholecalciferol) at a dose of 2000 IU per day and omega-3 fatty acids at a dose of 1g per day for prevention of cancer and CVD in 25,871 participants, which included men who were 50 years of age or older and women who were 55 years of age or older. The study included 5,106 black participants and the groups were balanced according to their gender. Standard questionnaires were administered to collect baseline data.- Inclusion Criteria: Absence of any history of cancer or CVD.
- Exclusion Criteria: These included renal failure or dialysis, liver cirrhosis, history of hypercalcemia, or any other serious conditions.
- Primary Endpoints: These include any type of invasive cancer and major cardiovascular events (composite of myocardial infarction, stroke, or death).
- Secondary Endpoints: Secondary cancer endpoints included colorectal, breast, and prostate cancers, and death from cancer. Secondary cardiovascular endpoints included an expanded composite of major cardiovascular events plus coronary revascularization and the individual components of major cardiovascular events.
Study Findings
The 25,871 participants were randomized to their respective groups from November 2011 through March 2014. The trial intervention ended on December 31, 2017, after a median follow-up of 5.3 years. The study population included 51% women. The mean age of the participants was 67.1 years. The cohort included 71% non-Hispanic white participants and 20% black participants; the rest were of other racial or ethnic groups.- Vitamin D: Baseline serum total 25-hydroxyvitamin D level was 31 ng/ml, which varied with age, sex, race or ethnic group, and BMI. Repeat estimation after 1 year revealed that 25-hydroxyvitamin D levels increased to 42 ng/ml in the vitamin D group. However, minimal change was noted in the placebo group.
- Cancer: The primary end point of invasive cancer of any type developed in 1,617 participants (793 in the vitamin D group and 824 in the placebo group). There were no significant differences between the two groups with regard to the incidence of breast, prostate, or colorectal cancer. During follow-up, 341 participants died from cancer (154 in the vitamin D group and 187 in the placebo group).
- Cardiovascular Disease: There were 805 major cardiovascular events during follow-up, which included myocardial infarction, stroke, or cardiovascular death (396 in the vitamin D group and 409 in the placebo group). Vitamin D supplementation also did not affect the risk of secondary cardiovascular endpoints.
- All-Cause Mortality: There were 978 deaths from any cause. The numbers of these deaths were not significantly different in the vitamin D group and the placebo group (485 and 493 deaths, respectively).
Conclusion
The incidence of cancer or major cardiovascular events in a US population was not reduced by daily supplementation with high-dose vitamin D for 5 years.Source of Funding
The study was funded by the National Cancer Institute, the National Heart, Lung, and Blood Institute, the Office of Dietary Supplements, the National Institute of Neurological Disorders and Stroke, and the National Center for Complementary and Integrative Health.Reference:
- Vitamin D supplements and prevention of cancer and cardiovascular disease - (https://www.nejm.org/doi/full/10.1056/NEJMoa1809944)
Source-Medindia
 MEDINDIA
MEDINDIA



 Email
Email