The versatility of the new method allows modification of peptides and could enable the development of peptide based drugs to treat a broad range of illnesses.

TOP INSIGHT
Researchers have developed a platform that could revolutionize peptide therapeutics. The cell-penetrating polymer brush can be attached to either of two specific amino acids, arginine or lysine, in the peptide chain.
Nathan Gianneschi, assistant professor of chemistry and biochemistry, guided the effort. Previous study by the group has revealed that peptides can be protected from digestion by arranging them as a densely packaged brush. The brush, a densely branching polymer, presents an overall, highly charged set of amino acids to the environment surrounding these molecules, which helps them to get inside cells.
Many molecules that stud the surfaces of cells hold a negative charge. The tip of one amino acid, arginine, holds a small positive charge. The polymer brushes chain multiple copies of this particular group of atoms, boosting the positive charge. The exact means of entry into the cells are not fully understood, but the researchers believe the charged brush helps the membrane engulf the peptide.
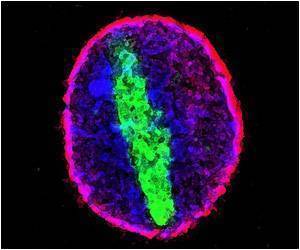
The research group worked out their system on a 'nonsense' peptide that has no known function in cells, but could be manipulated to attach varied appendages at different positions along the amino acid chain. The team then tested their strategy on a therapeutic peptide called KLA, which could prove useful for fighting cancer because it causes cells to self-destruct, but is unable to enter cells on its own. Once incorporated into a dense brush, KLA was able to enter cells and retained its killing power.
The researchers said, "The chemical procedure required to alter the peptide to create a cell-penetrating polymer brush is relatively simple. The brushes can be attached to either of two specific amino acids, arginine or lysine, in the peptide chain, and either could also be added to the end of a peptide that is lacking these attachment points."
Source-Eurekalert
 MEDINDIA
MEDINDIA



 Email
Email






