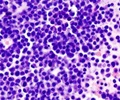
The first mouse model for the most common form of infant leukemia has been created by researchers.

TOP INSIGHT
The newly developed mouse model of infant leukemia replicates the human genetic flaw that causes this disease, making it much easier to study.
Children and adults with this disease produce vast numbers of dysfunctional blood cells, which eventually crowd out functional cells. MLL-AF4 leukemia has a dismal prognosis, among the worst of any subset of acute leukemia.
"For 20 years, scientists have repeatedly tried and consistently failed to make a model of MLL-AF4 Pro-B acute lymphoblastic leukemia," said Michael Thirman, Associate Professor of Medicine at the University of Chicago. "Even though we understood the basic genetic flaw, no one had been able create a mouse model that mimicked the human disease, which is crucial for evaluating potential therapies."
That frustrated many researchers, who shifted their focus to test alternative hypotheses on the causes of this leukemia or refocused their laboratories to study different aspects of this disease.
Thirman's team, including longtime colleague Roger Luo began working on this problem "years ago," he said, and stayed with it. They quickly identified two hurdles.
"We soon discovered that the virus wasn't working," Thirman explained. "We knew that certain parts of human DNA can decrease viral titers. So we switched from the human version of AF4 to the mouse version, Af4, which is slightly different. This increased viral titers 30 fold."
Next, they collaborated with James Mulloy at Cincinnati Children's Hospital Medical Center, whose graduate student Shan Lin inserted the fused MLL-Af4 gene into human CD34 cells, derived from cord or peripheral blood from volunteer donors. They transferred those cells to mice with immune systems that permit the growth of human cells. This time, the mice developed Pro-B ALL, identical to the leukemia found in humans.
"The model worked perfectly," Thirman said. Within 22 weeks, all of the mice developed exactly the same type of leukemia as observed in patients.
Expression of MLL-Af4 in human cells "recapitulates the pro-B ALL observed in patient with t(4:11) as shown by immunophenotype, chromatin targeting of the fusion, nuclear complex formation, and gene expression signatures," the authors wrote. "It mimics the disease found in humans both phenotypically and molecularly."
"The differences in the type of leukemia that developed using mouse versus human cells were striking," said Mulloy. "Researchers need to consider these differences carefully when choosing which model to use to mimic human disease. The available evidence now indicates that the approaches are not equivalent."
They conclude that "our MLL-Af4 model will be a valuable tool to study this most prevalent MLL-fusion leukemia with such a poor prognosis."
However, there is more work to be done. "MLL fusion disease is not a single genetic entity," the authors note. "Each has its own genetic and biological features associated with particular fusion partners." This highlights the need for "more models specific to each fusion. Our MLL-Af4 model will be a valuable tool."
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email