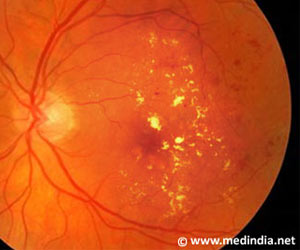
Vision loss associated with retinitis pigmentosa can be slowed dramatically by reprogramming the metabolism of photoreceptors, or light sensors, in the retina.

‘Vision loss associated with a form of retinitis pigmentosa can be slowed dramatically by reprogramming the metabolism of photoreceptors, or light sensors, in the retina.’





Columbia University Medical Center (CUMC) researchers have
demonstrated that vision loss associated with a form of retinitis
pigmentosa can be slowed dramatically by reprogramming the
metabolism of photoreceptors, or light sensors, in the retina. The study, conducted in mice, represents a novel approach to the treatment of RP, in which the therapy aims to correct downstream metabolic aberrations of the disease rather than the underlying genetic defect. The findings were published online today in the Journal of Clinical Investigation.
"Although gene therapy has shown promise in RP, it is complicated by the fact that defects in 67 genes have been linked to the disorder, and each genetic defect would require a different therapy," said study leader Stephen H. Tsang, the László Z. Bitó Associate Professor of Ophthalmology, Pathology and Cell Biology, and the Institute of Human Nutrition. "Our study shows that precision metabolic reprogramming can improve the survival and function of affected rods and cones in at least one type of RP. Since many, if not most, forms of the disorder have the same metabolic error, precision reprogramming could conceivably be applied to a wide range of RP patients."
Rods are among the most metabolically active cells in the body. They are particularly active during periods of darkness, when they burn glucose to release energy. In an earlier paper, Dr. Tsang and his colleagues theorized that rods deteriorate in RP, in part, because they lose the daytime's ability to use glucose to rebuild the rods' outer segment (the light-absorbing portion of the photoreceptor). "We hypothesized that diseased rods could be rescued by reprogramming sugar metabolism," said Dr. Tsang.
Dr. Tsang tested this hypothesis in mice with a mutation in the Pde6 gene that disrupts rod metabolism, leading to an RP-like disorder. The mice were treated so that their rods could not express Sirt6, a gene that inhibits sugar metabolism.
Advertisement
While the treatment significantly prolonged survival of the diseased rods and cones, it did not prevent their eventual death. "Our next challenge is to figure out how to extend the therapeutic effect of Sirt6 inhibition," said Dr. Tsang.
Advertisement
Dr. Tsang noted, "Further studies are needed to explore the exciting possibility that precision metabolic reprogramming may be used to treat other forms of RP and retinal degeneration."
Source-Eurekalert