Novartis’s drug, Gilenya used to treat multiple sclerosis has been be linked to two cases of rare infection called progressive multifocal leukoencephalopathy.
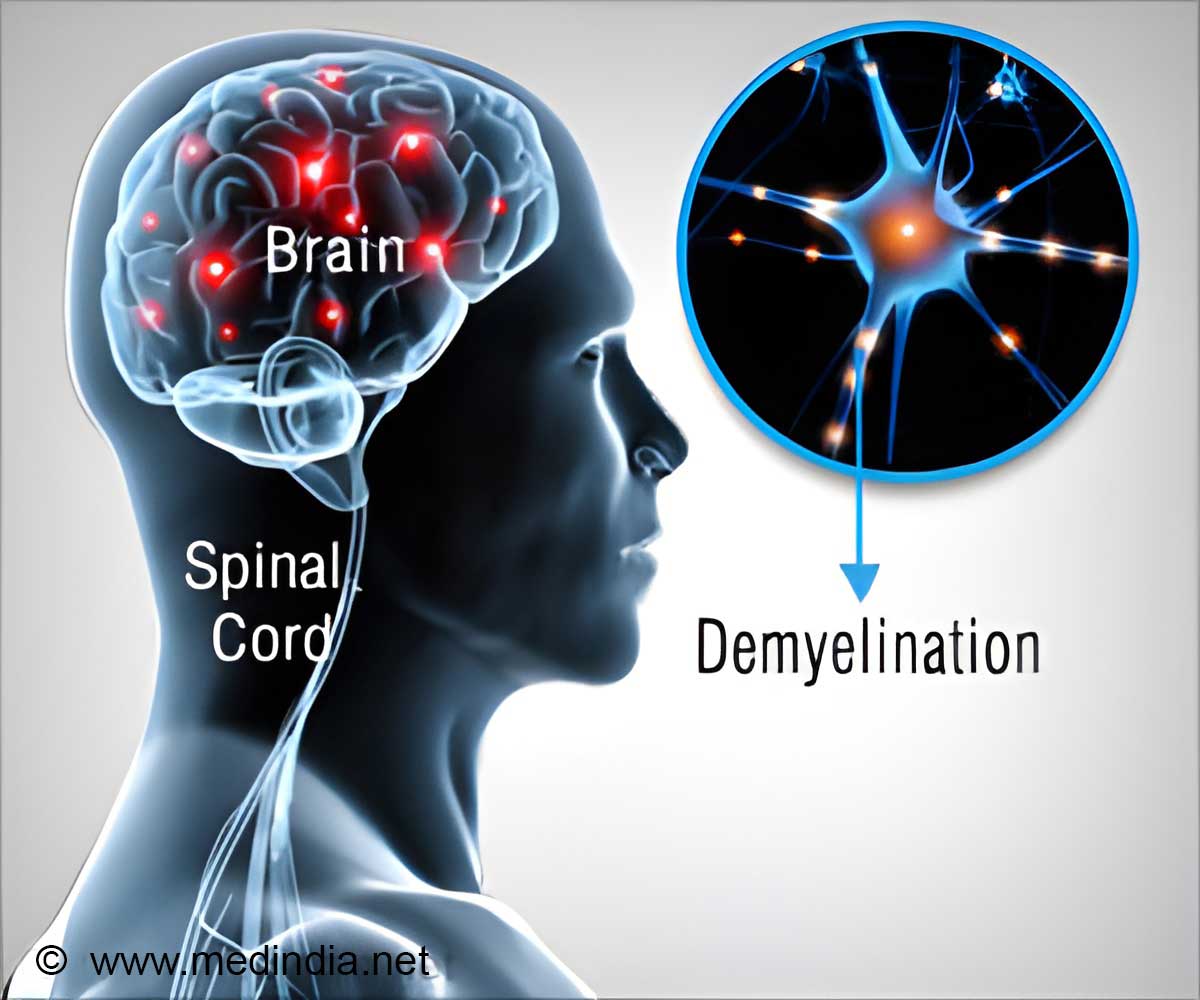
According to National Multiple Sclerosis Society, more than 2.3 million people are affected by Multiple Sclerosis, a chronic disease of the central nervous system.
Gilenya helps to reduce relapses and delayed disability progression in MS patients. The drug has been prescribed to about 54,000 U.S. patients from its approval in 2010 through July 2015, said the FDA.
Symptoms of PML include worsening weakness, motor problems in arms and legs, and changes in eyesight and thinking. Therefore, FDA is advising patients receiving Gilenya to consult their health care professionals if they experience these symptoms. Patients should not stop Gilenya treatment without first discussing it with their health care professionals.
Both health care professionals and patients are encouraged to report adverse side effects to the FDA’s MedWatch Safety Information Program.
In august 2013, a case of PML infection was reported to the agency. But it could not be directly linked with Gilenya, as the patient had been previously treated with an immunosuppressant drug that can cause PML.
Source-Medindia
 MEDINDIA
MEDINDIA



 Email
Email