Current therapy for stroke is unfortunately very limited. There is only one drug, tPA, and tPA needs to be administered within hours of onset of a stroke.

TOP INSIGHT
Despite public education and the development of stroke centers, only about 7 percent of all ischemic stroke patients nationwide receive tPA therapy.
Cathy Sila, MD, Director of the Comprehensive Stroke Center at UH Case Medical Center, and Professor of Neurology at Case Western Reserve University, was the principal investigator of the study at UH Case Medical Center.
"These results are very promising and this therapy would be an important adjunct to acute stroke care to reduce the amount of brain injury from which patients need to recover," said Dr. Sila. "But the results are still preliminary and need to be reproduced." Since MultiStem therapy seems to be effective when given at 24 to 36 hours after the stroke, it means many more stroke victims would be eligible for treatment, she said.
"Current therapy for stroke is unfortunately very limited. There is only one drug, tPA, that is FDA approved for acute ischemic stroke, and tPA needs to be administered within hours of onset of a stroke," said Dr. Sila. "Despite public education and the development of stroke centers, only about 7 percent of all ischemic stroke patients nationwide receive tPA therapy and less than 2 percent undergo catheter-based clot removal therapy. New treatments are needed to reverse the effects of a stroke and promote recovery from stroke, and they need to be effective in a wider time window to help more patients."
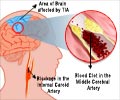
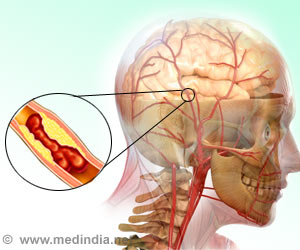
Ischemic stroke is caused by blockage in an artery in or to the brain, that impedes blood flow, and that can result in serious disability or even death. MultiStem® is a proprietary medication made by the Cleveland-based biotech company Athersys.
MultiStem® cells appear to reduce the local inflammatory response and protect neurons in the brain, while modulating the body's general immune response and inflammation which leads to additional damage to the brain in the days immediately following the stroke. This is an entirely new concept for how cell therapies may provide benefit following central nervous system injury.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email