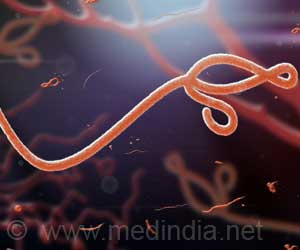
A new study has revealed that human antibodies can neutralize the Marburg virus, a close cousin to Ebola virus.
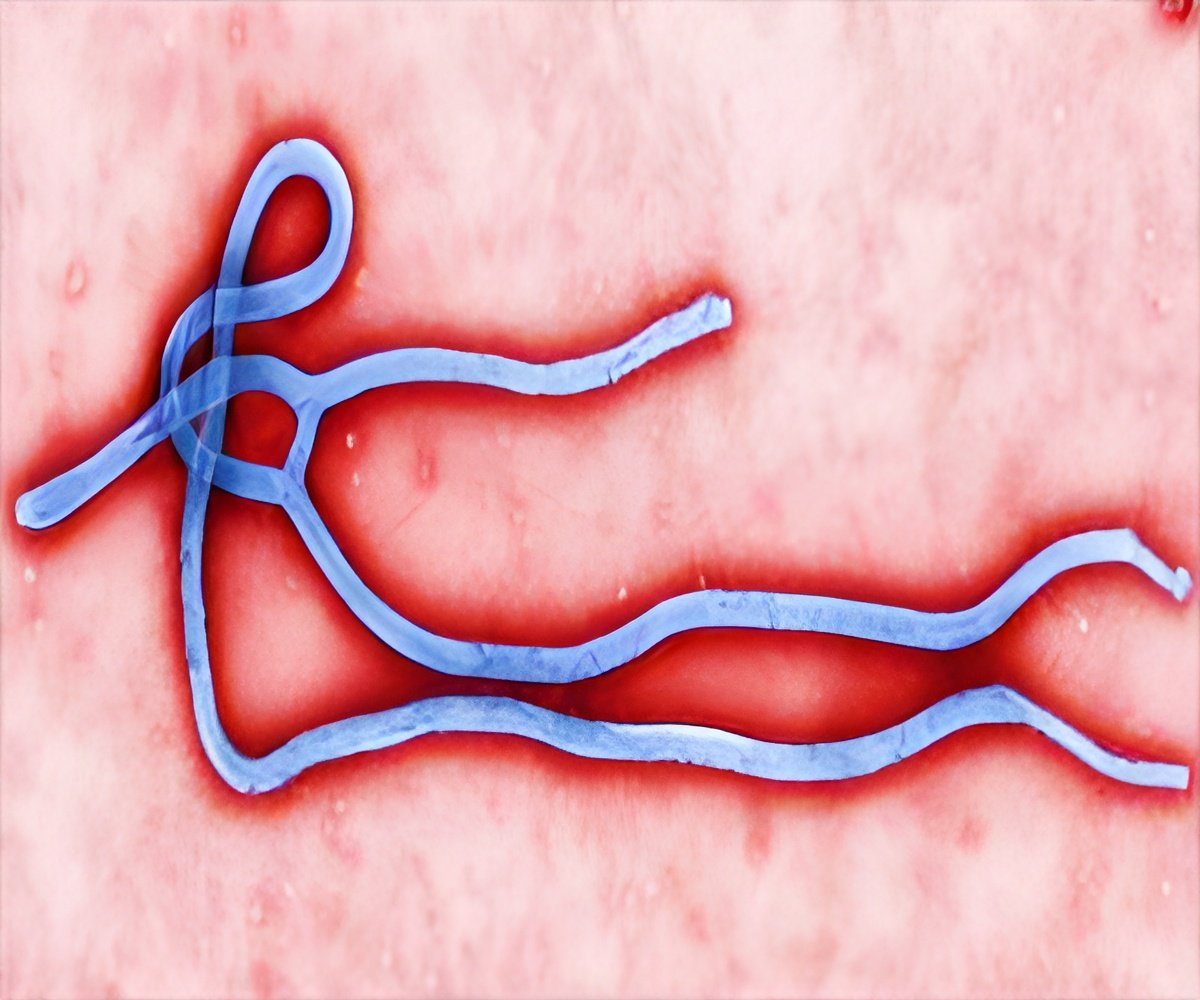
Crowe, Ann Scott Carell Professor and director of the Vanderbilt Vaccine Center, said, "The good news is, humans do make antibodies when they are infected that can kill these viruses, which suggests that vaccines should work." Researchers at Crowe's laboratory used a high-efficiency method to isolate for the first time human antibodies that bound a protein on the surface of the Marburg virus, in a woman's blood sample who was diagnosed with Marburg virus infection after returning to the United States from Uganda, where she had visited a fruit bat cave.
These findings were confirmed in the level 4 biosafety facility at UTMB's Galveston National Laboratory. No live virus was used in the research at Vanderbilt.
Researchers at The Scripps Research Institute in La Jolla, California, used X-ray crystallography methods to solve, for the first time, the structure of the Marburg virus bound to antibodies provided by the Crowe lab.
Vanderbilt is partnering with companies, including Mapp Biopharmaceutical Inc. in San Diego, California, to produce large quantities of Marburg and Ebola antibodies that could be used to provide short-term protection from the infection, especially for health care workers and family members who may be exposed to the viruses. Researchers are hoping that the clinical safety and dosage trials could be completed by the end of the year.
The study is published in the journal 'Cell'.
 MEDINDIA
MEDINDIA

 Email
Email