Hydrogels are a 3D network composed of hydrophilic polymer and have excellent swelling property which can absorb ten to thousand times of its dry weight in water.

TOP INSIGHT
By examining the behavior of sea anemone, experts found that mechanically durable protein in its body could play an important role in contraction and relaxation.
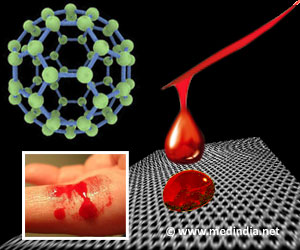
Based on the team's finding, the sea anemone-derived DNA was inserted in E. coli system for mass production because E. coli grows fast and produces protein better than sea anemone. This recombinant protein is named aneroin and is composed of amino acids, and among them, tyrosine is one of the rich amino acids in aneroin. Using abundant tyrosine residues, tyrosine-tyrosine (dityrosine) interaction was promoted for a mechanically improved hydrogel because dityrosine-linkage contributes to durable structures in nature including the jumping pad of dragonflies and fertilization membranes of sea urchin.
Through a photo-initiated dityrosine crosslinking method, the aneroin solution was transformed into a 3D hydrogel-based scaffold in a few seconds. Mechanically, the aneroin hydrogel exhibited significantly stronger and stiffer properties than those of collagen, gelatin, and elastin, which have already been widely exploited as hydrogel materials. It also exhibited approximately 4-fold stronger mechanical properties compared with silkworm silk.
Biologically, the aneroin hydrogel provided an adequate environment for cell growth. Mammalian cells inside the hydrogel proliferated well with an appropriate cell size and healthy morphology. Dead cells were barely detectable in the hydrogel. The mechanically durable and biologically favorable aneroin hydrogel shows clear advantages and could be used in various biomedical applications, especially for cell-containing biomaterials, cell-carrier patches, bio-artificial grafts, and burn dressing materials.
Source-Eurekalert
 MEDINDIA
MEDINDIA



 Email
Email







