Defibrillators – devices designed to detect and correct dangerous heart rhythms – can help people with heart disease live longer, and with a much better quality of life, suggests a study.

The MADIT-RIT trial, published today in the New England Journal of Medicine and presented at the American Heart Association's Scientific Sessions in Los Angeles, was led by Professor Arthur J. Moss, M.D., a world-renowned expert in the treatment and prevention of cardiac arrhythmias and sudden cardiac death. Moss' team found that simply raising the heart rate at which the device is set to fire – deliver therapy – made all the difference.
"The way we've been using implantable defibrillators for the last 20 years has really been less than optimal," said Moss, longtime professor of Cardiology at the University of Rochester Medical Center. "The extent to which the new programming reduced death and inappropriate therapies was quite striking and has the potential to beneficially affect a wide spectrum of patients who are at risk for sudden death or rhythm disorders."
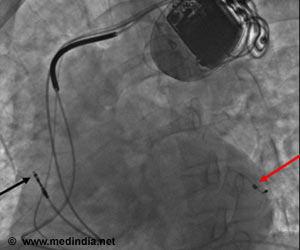
Implantable cardioverter defibrillators or ICDs constantly monitor the rate and rhythm of the heart and are supposed to deliver electrical shocks in response to very fast and potentially fatal heart rhythms. In 2002, Moss and the MADIT (Multicenter Automatic Defibrillator Implantation Trial) research group showed that ICDs are extremely effective in preventing death in patients at risk of irregular heart rhythms and sudden cardiac death, including individuals who've suffered a heart attack. The work changed medical guidelines nationwide, making thousands of heart attack survivors eligible for ICD therapy. Currently, around 200,000 ICDs are implanted in the U.S. every year.
But, according to a 2008 study in the Journal of the American College of Cardiology, approximately 20 to 25 percent of defibrillator therapy is inappropriate, meaning shocks are delivered in response to less dangerous rhythms that aren't likely to pose any immediate danger to patients. Such shocks are not only painful, but can take an emotional toll as well. Data from a 2002 trial comparing anti-arrhythmic drugs and defibrillators suggested that shocks are associated with reduced mental well-being. And a recent review of more than 45 studies found that such emotional distress is not uncommon, reporting that between 11 and 28 percent of ICD patients had some form of depression and between 11 and 26 percent had an anxiety disorder.
Moss' team conducted the MADIT-RIT (Reduce Inappropriate Therapy) trial, which was sponsored by Boston Scientific, to determine if different ways of setting the device – a complex process performed by a heart rhythm specialist prior to device implantation in a patient's chest – could reduce the occurrence of inappropriate therapy. Boston Scientific develops and markets medical devices, including ICDs and cardiac resynchronization therapy defibrillators or CRT-Ds.
Advertisement
Currently, most defibrillators are set to initiate therapy when the heart rate exceeds around 170 beats per minute, but rates of 180 or 190 are not always dangerous, are usually short-lived, and could be related to increased activity or exercise. Unfortunately, Moss says, defibrillators aren't very good at differentiating benign from malignant rhythms in this "in-between" range.
Advertisement
"There is considerable research to suggest that there is a small amount of damage to the heart muscle with each delivered shock," added Moss. "If we can eliminate the unnecessary shocks, this is going to be associated with less heart damage and improved outcomes."
The MADIT-RIT trial is the first large-scale, randomized study designed to evaluate specific programming features to reduce inappropriate therapy in patients with ICDs. The trial's sponsor, Boston Scientific, was not involved in data collection or data analysis. Moss, who has led the MADIT trials since their inception in the 1990s, holds no stock in any device company, has never been a member of any corporate speakers bureau, and since Dec. 1, 2008, has chosen not to accept honoraria from Boston Scientific for any professional activity.
In addition to Moss, Wojciech Zareba, M.D., Ph.D., Mary Brown, David Huang, M.D., Helmut Klein, M.D., and Scott McNitt from the University of Rochester Medical Center participated in the research. Researchers and physicians from Henry Ford Hospital, Hospital of the Good Samaritan, Duke University Medical Center, Tufts-New England Medical Center, Columbia University, Institute for Clinical and Experimental Medicine, University of Iowa Health Care, Tokyo Women's Medical University, and Loyola University Medical Center also contributed to the research.
Moss, who's spent a large part of his career working to improve ICD therapy, concludes, "When the findings were first uncovered there was a sincere, general enthusiasm that we had finally come to improve the therapy we've been using for 20 years, and that we've made true progress in making ICD therapy safer, more effective, and more acceptable to patients."
Source-Eurekalert