Oral-delayed release methylene blue aids in finding cancerous and pre-cancerous lesions, found study.
TOP INSIGHT
Oral-delayed release methylene blue aids in finding cancerous and pre-cancerous lesions.
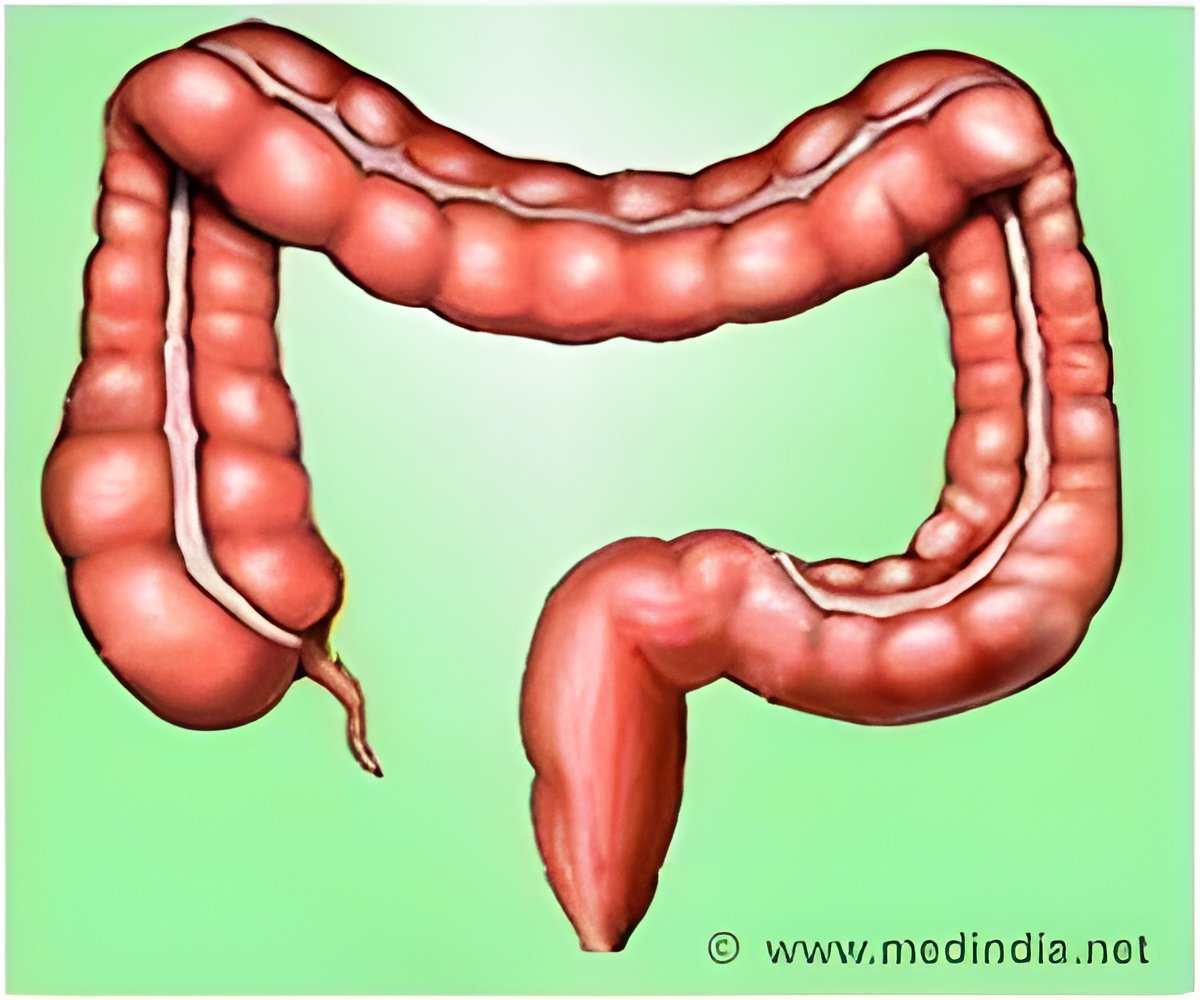
Every year, nearly 137,000 people are diagnosed with CRC, and more than 50,000 people die from the disease. Yet, the disease is largely preventable with regular screening and is treatable with early detection. The challenge is that polyps are not always detected during screening -- many polyps are flat or subtle, making them difficult to identify and remove. "Identification of cancerous and pre-cancerous lesions is of utmost importance to prevent CRC," said Alessandro Repici, MD, professor of gastroenterology and director of endoscopy at Humanitas University Medical School in Milan, Italy, and a primary investigator of the study. "Our study, which used the highest standard of care, allowed gastroenterologists to better detect and remove difficult-to-see polyps, which has great implications for further preventing this disease."
Study design
Researchers studied 1,205 patients scheduled for colonoscopy at 20 centers worldwide, with each patient randomly assigned to one of three groups: patients who received a full dose of the blue dye, oral delayed-release methylene blue, during the normal colonoscopy preparation process; patients who received a placebo during preparation; and a group of patients who received a half dose of the study drug. The third group was not part of the analysis but was included for masking purposes to make it harder for participating physicians to know which patients were in the active group.
Study results
Previously, the blue dye had to be mixed by the providers on site, and then sprayed during the colonoscopy, which could be an imprecise, time-consuming and generally localized process. With the development of the tablet form, the majority of the dye releases in the colon in time for highlighting and detecting mucosal lesions during the colonoscopy. Study investigators added that the use of Methylene Blue MMX or other technologies should never be considered a substitute for good colonoscopy technique.
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email






