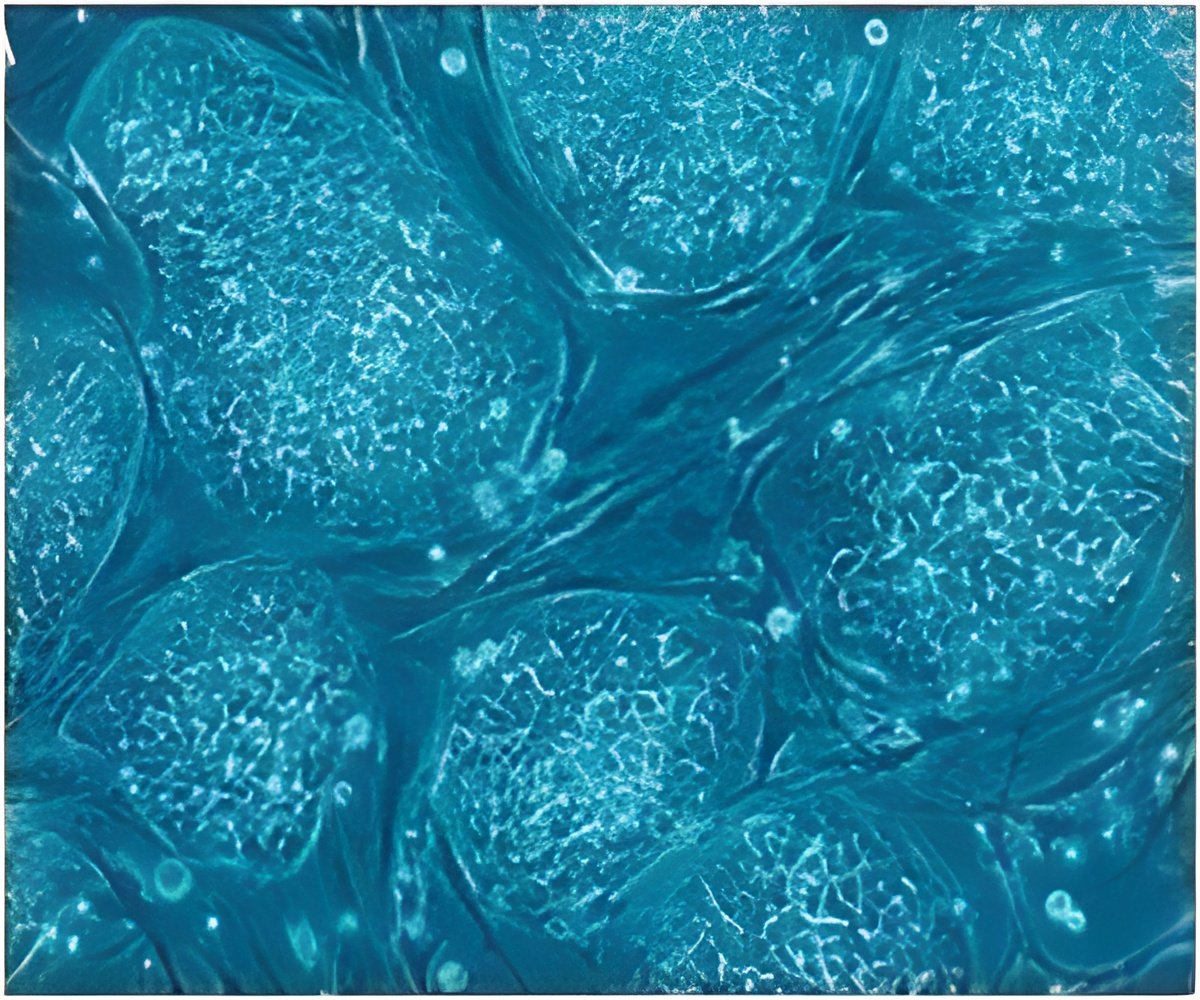
Histone BigH1 is the key protein that causes the differentiation of stem cell to male sex cells.
TOP INSIGHT
Histone BigH1 is either present and represses specific genes or is inhibited to allow gene expression, thereby promoting differentiation, a process in which stem cells produce adult sex cells.
The role of histone H1 in male infertility
All animals have histones that are specific to the germ line. In Drosophila, BigH1 is expressed in the male and female germ lines. In contrast, variants of histone H1 differ between males and females in mice and in humans. "The histone BigH1, which we have studied in Drosophila melanogaster, shows most resemblance to the germ line variant of human females but it also shows some similarities to variants of the male germ line," explains Albert Carbonell, postdoctoral fellow and first author of the paper. "Because of these similarities, our work can contribute to our understanding of sex cell diferentiation in humans and help to explain some types of infertility," he adds.
Histones as regulators in stem cell differentiation into tissue cells
All the cells of an organism, whether skin, intestinal, lung or any other type, arise from the differentiation of stem cells, and it is known that histones play a crucial role in this process in many lineages. This study describes the mechanism of action and inhibition exerted by BigH1 to regulate spermatogenesis in Drosophila and which may work in a similar manner in other species.
The lab is now focusing on BigH1 during the differentiation of female sex cells. "We know that this histone is essential, but we assume it will have a distinct role since the process involving female sex cells is considerably different to that of male ones," says Paula Climent, a PhD student in the lab and one of the authors of the article.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email










