Atezolizumab marketed as Tecentriq has shrunk bladder tumors and expanded the survival of these patients.
TOP INSIGHT
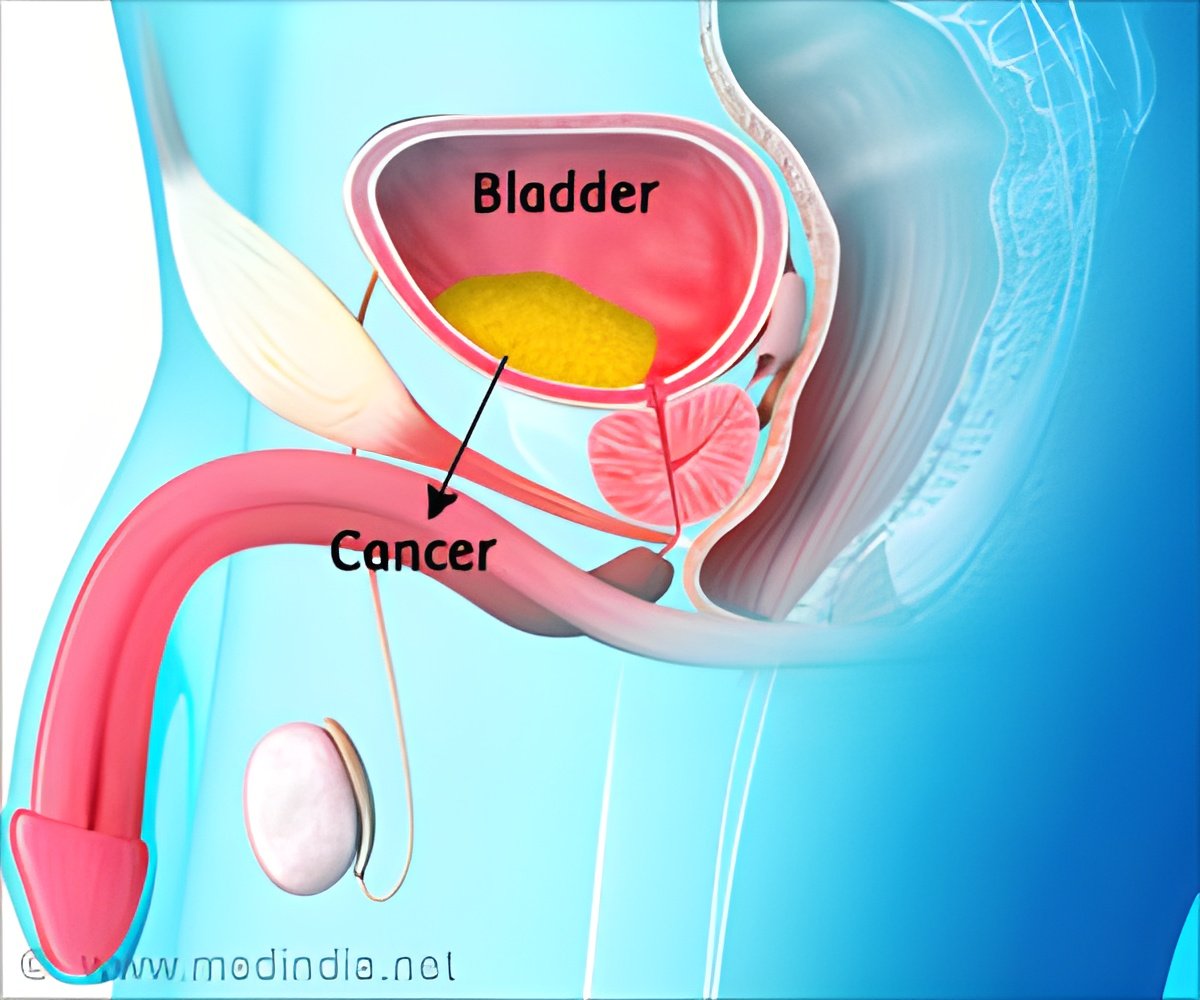
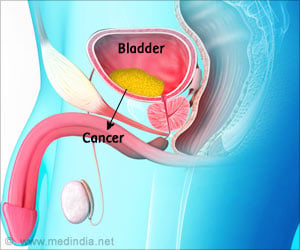
Immunotherapy drug Atezolizumab marketed as Tecentriq has shown potential to shrink tumors and expand survival rates of people with advanced bladder cancer.
Also, these patients were reported to be in remission. Their average overall survival was just under 15 months. About 10 to 15 percent of patients experienced severe side effects from Tecentriq such as hypothyroidism, liver problems and diarrhea.
The drug acts on the immune system and activates monoclonal antibodies, responsible for destroying tumors. These antibodies inhibit a specific genetic interaction that allows cancer cells to evade the immune cells.
The results were found to be slightly lesser effective than the currently available platinum-based chemotherapy for bladder cancer. USFDA approved atezolizumab drug for bladder cancer in May 2016.
"Immunotherapy really treats your body's immune system so your immune system can fight the cancer for you," said Dr. Arjun Balar, an assistant professor at NYU Langone Medical Center who treated some of the patients.
Source-Medindia
 MEDINDIA
MEDINDIA

 Email
Email