Topotecan Medication Information
Learn everything you need to know about Topotecan-pronunciation, uses, dosage guidelines, indications, and when to take or avoid it.
Get up-to-date information on side effects, precautions, warnings, and proper storage to ensure safe usage.
Explore Topotecan brand names commonly used in India and internationally, along with detailed pricing information. Consult your healthcare provider for tailored medical advice.
Generic Name : Topotecan Pronunciation : toe'' poe tee' kanBrand Names or Trade Names of Topotecan
India :
International :
Hycamtin
Why is Topotecan Prescribed? (Indications)
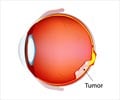
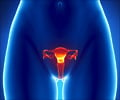
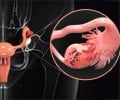
This medication is a topoisomerase type I inhibitor, prescribed for ovarian or lung cancer that do not respond well to other types of cancer treatment. It kills the cancer cells in ovary and lungs.When should Topotecan not be taken? (Contraindications)
Contraindicated in pregnant and breastfeeding women, and hypersensitivity.What is the dosage of Topotecan?
IV- The recommended dose is 1.5 mg/m2/day over 30 minutes, on 1 to 5 days. It is given in uterus and small cell lung cancer as an IV infusion in a dose of 1.5 mg/m2/day for the first 5 days of a 21-day course. The patient has to undergo at least 4 courses.In cancer of the cervix a dose of 0.75 mg/m2 is given for 3 days along with cisplatin on the first day of a 21-day course.
How should Topotecan be taken?
This medication available in the form of capsule to take by mouth with a full glass (8 ounces) of water, with or without food. (Swallow whole, do not open/ chew/ crush.)Injection: Intravenously (into a vein) over 30 minutes by healthcare providers.
What are the warnings and precautions for Topotecan?
•Caution needed for patients with history of pre existing bone marrow depression.• Frequent monitoring of blood counts during treatment.
• It may cause dizziness, do not drive a car or operate machinery while taking this medication.
What are the side effects of Topotecan?
Central Nervous System- Fatigue, weakness, discomfort,headache.Blood- Abnormal low white blood cell count, hemoglobin.
Miscellaneous- GI upset, total hair loss, shortness of breath, itching and increased levels of bilirubin.
 MEDINDIA
MEDINDIA
 Email
Email