Avatrombopag Medication Information
Learn everything you need to know about Avatrombopag-pronunciation, uses, dosage guidelines, indications, and when to take or avoid it.
Get up-to-date information on side effects, precautions, warnings, and proper storage to ensure safe usage.
Explore Avatrombopag brand names commonly used in India and internationally, along with detailed pricing information. Consult your healthcare provider for tailored medical advice.
Generic Name : Avatrombopag Pronunciation : a-va-TROM-boe-PAG Therapeutic Classification : Blood Modifier AgentsBrand Names or Trade Names of Avatrombopag
India :
Doptelet
Overview of Avatrombopag
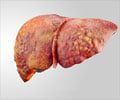
Avatrombopag tablets for oral use were approved by the FDA in May 2018 for treating thrombocytopenia in adults with severe liver disease and scheduled to undergo a medical or surgical procedure.Why is Avatrombopag Prescribed? (Indications)
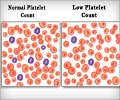
Avatrombopag tablets are prescribed for treating abnormally low platelet counts or a condition of thrombocytopenia in adult patients with severe liver dysfunction who are planning to undergo a medical or dental procedure.Thrombocytopenia is found to be a common complication in patients with severe liver disease either due to liver pathologies or as a result of interferon-based antiviral treatment.
Avatrombopag is a small molecule thrombopoietin (TPO) receptor agonist that works by rapidly increasing the number of platelets from the megakaryocytes in the bone marrow and thereby reducing the need for blood transfusions.
However, it does not result in increased platelet activation which could lead to blood clotting and several complications.
When should Avatrombopag not be taken? (Contraindications)
Avatrombopag should not be used with a focus to normalize platelet counts and in patients with-• Allergy to avatrombopag
• Pregnancy and breastfeeding
• Children below 18 years of age
• History of clotting disorders (e.g., Thrombophilia)
• Severely dehydrated patients (e.g., Hyponatremia or abnormally low sodium level)
Caution is required in treating the following patients as safety and effectiveness parameters have not been studied in these groups
• Severe kidney disease or in patients undergoing hemodialysis
• Geriatric cases
What is the dosage of Avatrombopag?
The recommended adult dose of avatrombopag depends on the individual patient’s platelet count performed before a procedure is planned.Platelet count (×109/L) and the recommended dose:
• Count 50 to 40 × 109/L: Take 40 mg (2 tablets) of avatrombopag once a day for five consecutive days
• Count below 40 × 109/L: Take 60 mg (3 tablets) of avatrombopag once daily for five consecutive days
How should Avatrombopag be taken?
• Avatrombopag tablets are available at a strength of 20 mg and should be taken by mouth once a day with food to avoid gastric discomfort.• Do not take for more than 5 days unless otherwise recommended by the physician.
What are the warnings and precautions for Avatrombopag?
• Platelet count should be performed before starting avatrombopag treatment and also repeated on the day of the procedure to assess the improvement in platelet count.• To protect the child from exposure to avatrombopag, breastfeeding mothers should not be allowed to breastfeed their child during the treatment and for at least two weeks after the last dose of avatrombopag.
• Patients should be closely monitored for any clotting risk or thromboembolic complications to prevent serious life-threatening or fatal conditions.
• Complete blood cell count and electrolyte evaluation should be performed regularly to detect the risk of low sodium level and other related blood disorders and treated accordingly.
What are the side effects of Avatrombopag?
The most common side effects experienced by the patients taking avatrombopag are-• Nausea
• Headache
• Fever
• Low sodium levels
• Stomach pain
• Fatigue or weakness
• Swelling of the extremities such as legs or hands
Other side effects include-
• Anemia
• Risk of developing blood clots
• Muscle pain or tenderness
• Difficulty in breathing
• Pain in the chest
• Abnormalities in heart rhythm
What are the other precautions for Avatrombopag?
• If a dose is missed, it should be taken immediately as soon as you remember.• Never take a double dose to make up for the missed dose.
• The dose of avatrombopag should not be stopped without the consent of the physician.
What are the Drug Interactions of Avatrombopag?
• Patients should not take anticoagulants such as warfarin or heparin as it may increase side effects of avatrombopag such as risk of blood clotting and other thrombotic or thromboembolic complications.• Diuretic drugs or water pills such as furosemide, hydrochlorothiazide or indapamide should not be taken with avatrombopag as it may cause severe electrolyte depletion such as sodium loss resulting in serious illnesses.
• Before starting avatrombopag therapy, the prescriber should be informed about intake of all prescription or non-prescription drugs or herbal supplements by the patients.
What are the storage conditions for Avatrombopag?
• Store avatrombopag tablets at room temperature between 20°C and 25°C.• Keep the tablets in its original package and protect from excess heat and moisture.
• Keep out of reach of children.
 MEDINDIA
MEDINDIA
 Email
Email