Micra pacemaker can be connected directly with the heart and has a 10-year battery life. It is also approved for full body MRI scans.
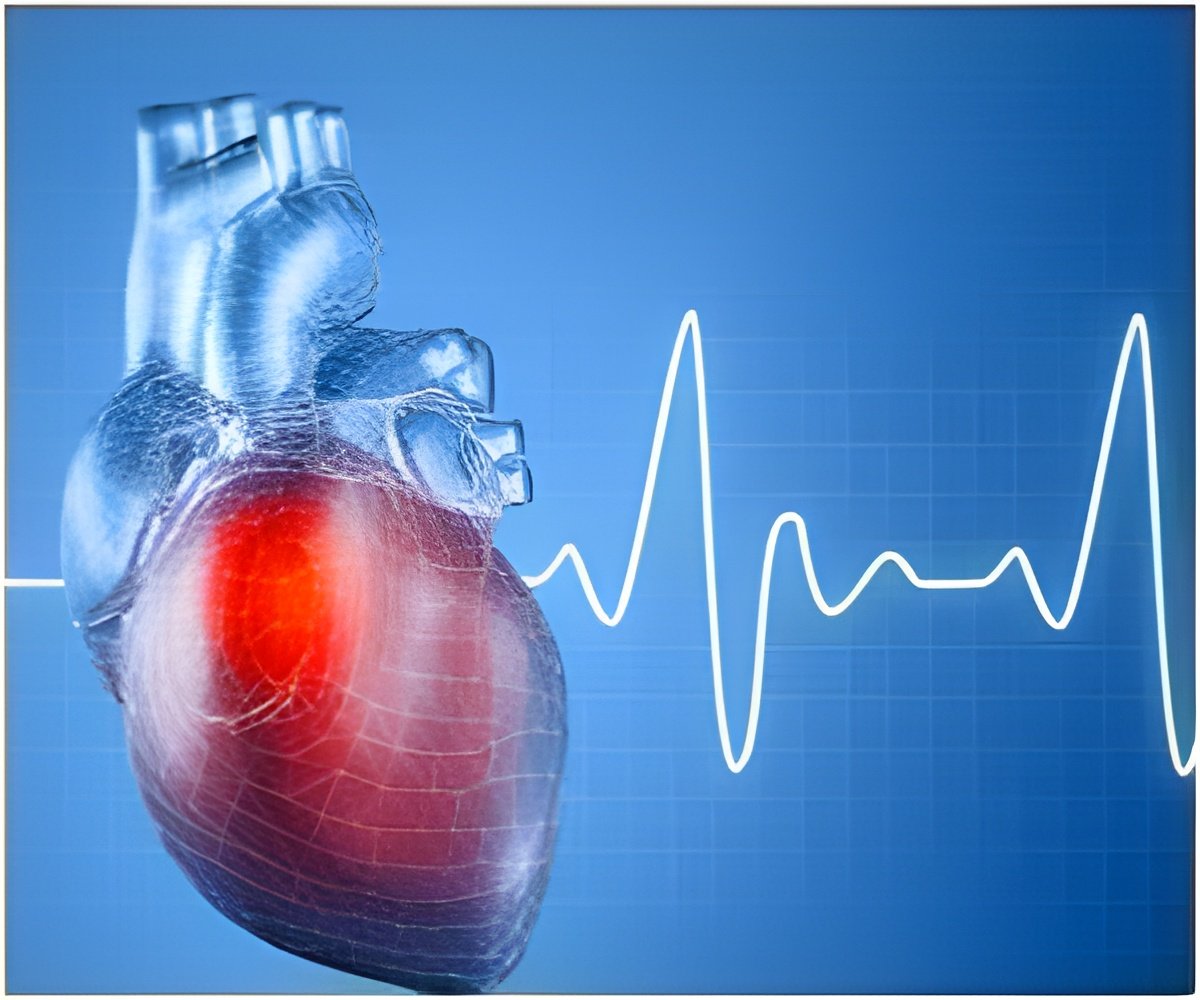
The chances of complications are more when a chest pocket for the pacemaker is created. Micra, does not need a pacemaker pocket. The device and the heart are attached directly using tines with the electrode at the tip delivering the pacing signals.
The Micra TPS has an estimated 10-year battery life despite its miniaturized size and is approved for full body MRI scans, providing patients with access to the most advanced imaging diagnostic procedures.
Based on results from the first 60 patients in the Medtronic Micra TPS Global Clinical Trial the device was awarded CE Mark. The trial is still ongoing and will continue to evaluate the safety of the device.
Source-Medindia
 MEDINDIA
MEDINDIA

 Email
Email







