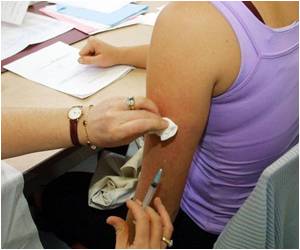
The World Health Organization (WHO) has approved administration of meningitis vaccine, which does not have to be stored in fridges or iceboxes, to babies across Africa.

The vaccine manufactured by Serum Institute of India was launched in 2010 and has been administered to more than 215 million people in the 15 countries of the African meningitis belt including Benin, Burkina Faso, Cameroon, Chad, Ethiopia, Gambia, Ghana, Ivory Coast, Mali, Niger, Mauritania, Nigeria, Senegal, Sudan and Togo.
Marie-Pierre Preziosi, the head of MVP, said, "Initial mass vaccination campaigns with MenAfriVac have been highly effective in reducing the number of meningitis A cases. Until now, however, the vaccine has only been administered in children and young adults between the ages of one and 29, and unprotected infants could undo the progress being made. Epidemics will return when rising numbers of unprotected newborns become a larger proportion of the total population over time. With this decision, health officials will be able to ensure that population-wide protection is sustained by routinely immunizing infants.”
Source-Medindia
 MEDINDIA
MEDINDIA


 Email
Email







