Taking a drug holiday from prolonged use of an osteoporosis drug group - bisphosphonates may result in fracture, finds a new study.

‘Patients at high risk of fracture who take drug holidays from bisphosphonates should be closely followed, especially if drug holiday becomes lengthier.’





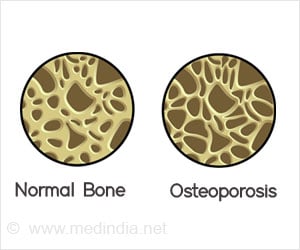
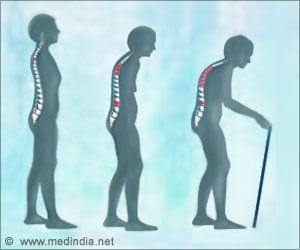
Patients at high risk of fracture who take drug holidays should be closely followed, especially as the drug holiday lengthens, researchers wrote.Bisphosphonates are the most common medications prescribed for osteoporosis. The drugs slow down the breakdown of bones, helping to maintain bone density and reduce the risk of fractures.
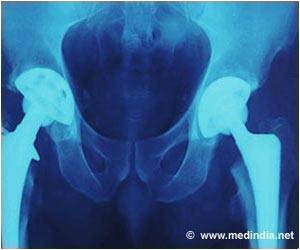
Bisphosphonates have been linked to osteonecrosis of the jaw (ONJ) and atypical femur fracture. ONJ occurs when the jawbone is exposed, typically following a dental procedure, and begins to weaken and die. An atypical femur fracture is an unusual fracture of the thigh bone that can occur even with normal weight bearing.
To reduce the risk of these side effects, the American Association of Clinical Endocrinologists and American College of Endocrinology recommend that women at moderate risk for osteoporosis take a drug holiday after five years of oral and three years of intravenous bisphosphonate treatment. Women at higher risk for osteoporosis should take a drug holiday after 10 years of oral and six years of intravenous bisphosphonate treatment.
However, there is minimal data on how long drug holidays should last. The Loyola study was designed to further characterize the increased fracture risk in patients taking drug holidays. The retrospective study examined the records of 371 women and 30 men with osteoporosis or osteopenia who began drug holidays. (Patients with osteopenia have weak bones, but not yet osteoporosis.) The patients had taken bisphosphonates for an average of 6.3 years before beginning drug holidays. The two most frequently prescribed bisphosphonates were alendronate (Fosamax®), taken by 62 percent of patients, and risedronate (Actonel®), taken by 34 percent of patients.
Advertisement
Drug holidays need further assessment, Dr. Camacho and colleagues wrote. "Patients who begin drug holidays at high risk for fracture based on bone mineral density, age or other clinical risk factors warrant close follow-up during the holiday, especially as its duration lengthens. Fracture risk needs to be regularly assessed during the drug holiday and treatment resumed accordingly."
Advertisement