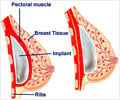
Experts in the UK say that tests on a type of breast implant filled with an unapproved gel have shown no evidence they are unsafe.
The Medicines and Healthcare products Regulatory Agency (MHRA) said in March that no more PIPs should be implanted and that any remaining should be returned to the manufacturer.
To determine how the altered version might react with the human body, the UK plastic surgeons organisation BAAPS (British Association of Aesthetic Plastic Surgeons) said French colleagues contacted the gel manufacturers for any studies.
UK tests of the implants have found no evidence of chemical toxicity or cancer risk from the gel.
"It's reassuring that our test results have shown no evidence of any associated risks with the filler material," the BBC quoted Susanne Ludgate, director of clinical devices for the agency as saying.
"We are however waiting for the results of the French tests which are more extensive and include mechanical testing of the implant shell because there maybe a suggestion of an increased rupture rate compared with other breast implants.
Advertisement
BAAPS said it was pleased the MHRA's findings had come out so quickly. Nigel Mercer of BAAPS, said: "Hopefully this will reduce the anxiety of patients whilst further studies are expected from France.
Advertisement
Source-ANI