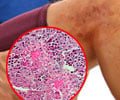
The immune mechanisms that lead to allergy and asthma have continued to puzzle researchers
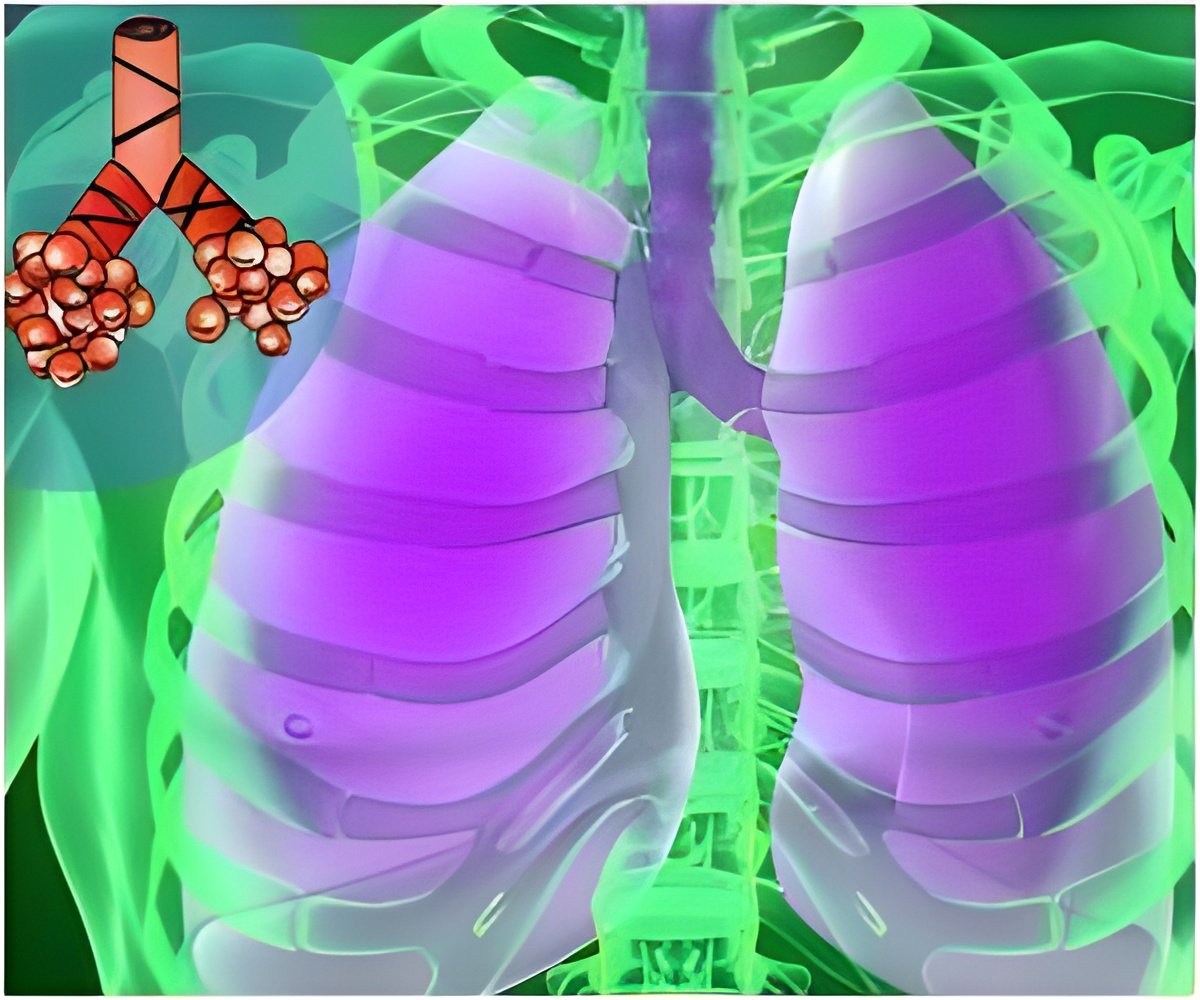
Asthma is part of a battle that takes place as the immune system marshals its forces to fight off an invading organism-or what mimics such invaders. The ensuing fight takes a significant toll on the human airway and lungs, often generating a violent and itself potentially deadly reaction – asthma.
In 2002, Corry and his colleagues found that proteinases, enzymes that chop up other proteins, are important in initiating the adaptive immune response that prompts generation of the critical T-cells and B-cells that populate the adaptive immune system. The adaptive immune system specifically targets allergens, and Corry knew that the more immediate innate immune system also played an important role in asthma and allergy.
"If you take many proteinases and give them to mice, they will induce an allergic disease that resembles asthma," he said.
With that key finding in the adaptive immune system, the researchers turned their attention to the puzzle presented by the innate immune system.
"What is the relationship between proteinases and asthma?" he said. Other work in the field pointed to another immune molecule called toll-like receptor 4 that was believed to play a role in activating T-helper type 2 (Th2) cells.
Advertisement
"Toll-like receptor 4 is not required for the Th2 response itself," said Corry. "But, the Th2 response is proteinase dependent."
Advertisement
In the airway, the same fibrinogen fragments that are part of the blood clotting process can cause clotting that is a barrier to breathing, said Corry.
In his studies, he used proteinase-producing fungi as the environmental trigger for asthma. Laboratory mice that lacked toll-like receptor 4 did not mount a robust allergic airway disease when challenged by proteinase, viable fungi or other triggers but did have a normal Th2 immunity.
"Why do our bodies do this?" said Corry. The answer is both simple and complicated. The system developed to allow organisms to survive infection with deadly organisms such as fungi. How it achieves that is complicated. In this "survival mode," the immune system generates symptoms that can themselves create disease.
Against the insidious onslaught of organisms such as fungi, which can kill if left unchecked, asthma may be a better alternative, said Corry.
"If you don't fight fungi off, they will get you," he said.
Source-Eurekalert