The growth and survival of prostate cancer cells are very dependent on signals that the cancer cells receive through a protein called the androgen receptor, said Daniel.
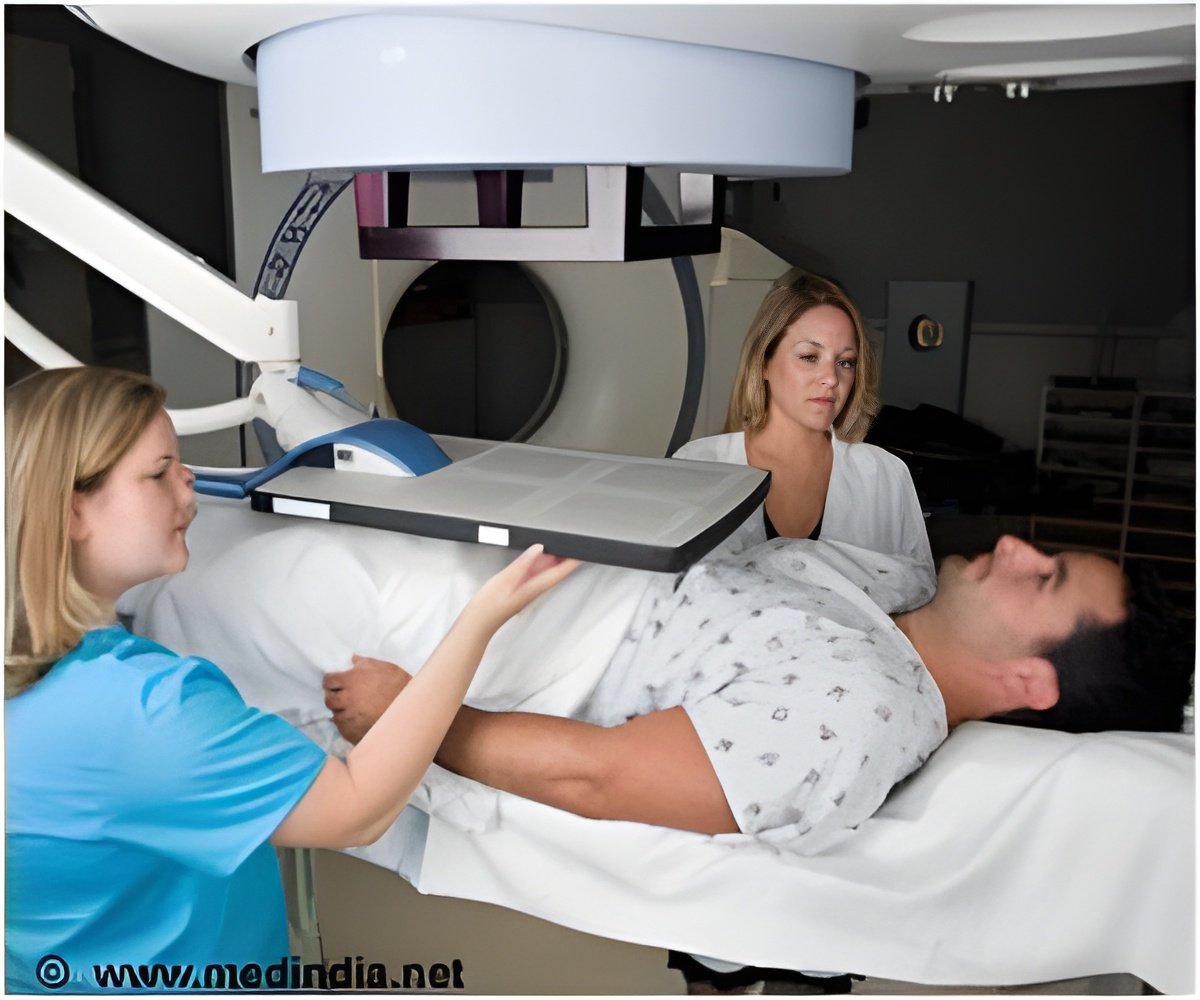
Haber and his colleagues established a way to isolate cancer cells from the blood of patients with prostate cancer and to measure readouts of androgen receptor signaling in each of the individual cancer cells in the blood.
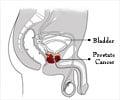
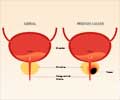
Deciding the ideal treatment for patients with metastatic prostate cancer that stops responding to initial therapy could be guided by certain analyses of cancer cells isolated from the patients'' blood, according to data published in Cancer Discovery, a journal of the American Association for Cancer Research.
Prior to the initiation of androgen-deprivation therapy, the androgen receptor signaling pathway was turned on in most of the cancer cells in the blood of patients with newly diagnosed metastatic prostate cancer. After the initiation of androgen-deprivation therapy, the pathway turned off in the circulating tumor cells.
However, in patients whose prostate cancer had progressed after initially responding to androgen-deprivation therapy, the cancer cells in the blood were highly variable. Some cells had the androgen receptor signaling pathway turned on while other cells had it turned off. Yet other cells had characteristics of the signaling pathway being both on and off. The presence of cells with a mixed androgen receptor signaling pattern was associated with an adverse treatment outcome.
In addition, in patients treated with a new drug, abiraterone, which achieves more complete androgen deprivation than earlier treatments, an increased percentage of circulating tumor cells with androgen receptor signaling turned on despite abiraterone treatment was associated with decreased overall survival.
"As more drugs are developed that target the different pathways that drive the recurrence of metastatic prostate cancer in different patients, it will become essential to know which drug and which pathway is relevant in each patient," he said. "Our assay will be an effective way to interrogate the tumor and follow it during the course of treatment to monitor therapy response and the emergence of drug resistance."
Source-Newswise
 MEDINDIA
MEDINDIA
![Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis Prostate Specific Antigen [PSA] & Prostate Cancer Diagnosis](https://images.medindia.net/patientinfo/120_100/prostate-specific-antigen.jpg)
 Email
Email