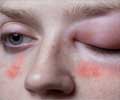
A C1-esterase inhibitor (human) has been approved by the FDA for the prevention of attacks of hereditary angioedema.
- Attacks of hereditary angioedema can be life-threatening, especially if they affect the tissues around the larynx
- A medication has been recently approved by the FDA to prevent such attacks in susceptible individuals
- Since it is administered subcutaneously, patients and their caregivers can be trained to administer it at home with ease
The affected individuals lack the functional form of a protein called C1-esterase inhibitor (C1-INH). C1-INH controls inflammation mediated by chemicals which constitute the ‘complement system’. In the absence of the protein, the inflammatory process goes unchecked, resulting in seepage of fluid from the blood vessels into the surrounding tissues and swelling of various tissues. The disease may be inherited from parents or may occur due to a new genetic mutation in the baby.
There are three types of hereditary angioedema, types 1, 2 and 3. Type 1 occurs due to deficient C1-INH. In type 2 hereditary angioedema, the C1-INH is present but dysfunctional C1-INH. The C1-INH in type 3 disease is normal, and the exact cause of the condition is not known.
Attacks of hereditary angioedema are treated with special drugs like recombinant human C1-INH, ecallantide and icatibant which arrest the inflammatory process. Drugs like danazole have been used for the prevention of hereditary angioedema. An intravenous C1-esterase inhibitor is also available but may be more inconvenient to use as compared to a subcutaneous preparation.
The recent approval of the subcutaneous C1 Esterase Inhibitor (Human) for the prevention of hereditary angioedema attacks comes following its successful use in the Phase III COMPACT trial. In this trial, the treatment was tested in patients with type 1 or type 2 hereditary angioedema between the ages of 12 and 72 years. Out of the 90 patients included in the study, some received the injection twice a week in doses of 40 IU/kg or 60 IU/kg for 16 weeks, while others received a placebo. The study concluded that:
- Patients in the group that received the medication experienced a considerable reduction in the number of attacks of hereditary angioedema as compared to those who received only placebo.
- The subcutaneous injections restored the level of C1-INH to more than 40%
- Adverse effects included allergic reactions, nasal and throat swelling and dizziness
- FDA approves first subcutaneous C1 Esterase Inhibitor to treat rare genetic disease - (https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm564332.htm)
- Longhurst H et al. Prevention of Hereditary Angioedema Attacks with a Subcutaneous C1 Inhibitor. N Engl J Med 2017; 376:1131-1140 DOI: 10.1056/NEJMoa1613627
Source-Medindia