The cardiovascular drugs sacubitril/valsartan and ivabradine have been included in recent guidelines by the ACC, AHA and the HFSA for the treatment of severe heart failure.
- Sacubitril/valsartan and ivabradine are drugs that help in the treatment of severe heart failure through different mechanisms.
- They reduce the need for hospitalizations due to heart failure
- They have been included by international cardiovascular organization in the guidelines for the treatment of severe heart failure.
Sacubitril acts by a different mechanism. It prevents the breakdown of natriuretic peptides by inhibiting an enzyme called neprilysin. Natriuretic peptides are substances that are naturally released by the body during heart failure and relax blood vessels and excrete salt and water. Thus, Sacubitril adds to the effect of valsartan in reducing the work load of the heart.
The combination should not be used in patients who had previously experienced angioedema (severe swelling) while taking an angiotensin converting enzyme (ACE) inhibitor or ARB, or due to any other cause. It should not be used along with aliskiren in patients with diabetes or an ACE inhibitor (ACE inhibitors should be stopped at least 36 hours before starting this combination). Adverse effects include low blood pressure, high blood potassium levels, cough, dizziness and kidney failure.
The beneficial effect of the combination was demonstrated in the PARADIGM-HF trial, which compared it to enalapril, an ACE inhibitor, in patients with an ejection fraction of less than 40% and a B-type natriuretic peptide level of between 100 and 150 pg/ml. The combination significantly reduced deaths due to cardiovascular causes and hospitalization due to heart failure. It also reduced the symptoms and physical limitations in patients due to heart failure.
The second drug discussed in the article was ivabradine. Ivabradine was approved by the US FDA in 2015 for patients with left ventricular heart failure with an ejection fraction of 35% or less, with a normal heart rate and rhythm, and are either on a maximum dose of beta blocker or cannot use a beta blocker. It acts on the sinoatrial node, the natural pacemaker of the heart, and slows down the heart. It should not be used in acute decompensated heart failure (sudden worsening of heart failure), severe liver dysfunction, in patients with an artificial cardiac pacemaker, a blood pressure <90/50 mmHg or a resting heart rate of less than 60 beats per minute. Some patients suffer from visual problems, which usually resolve with time, or after stopping the medication.
About Heart Failure
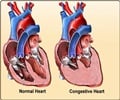
The heart acts as a pump whose continuous and rhythmic contractions and relaxations pump the blood throughout the body. A failure of the heart to carry out this function results in heart failure. Heart failure is classified at right-sided failure, when the right side of the heart is affected, and left-sided failure when the left side of the heart is affected. Left-sided heart failure or left ventricular dysfunction can be systolic dysfunction, where the contraction of the heart is affected, or diastolic dysfunction, where the heart fails to relax and thereby fill up properly. Patients with systolic dysfunction have reduced ejection fraction, while the ejection fraction is normal in those with diastolic dysfunction. The ejection fraction refers to the percentage of blood that is pumped out of the left side of the heart during its contraction. The normal ejection fraction is 55% or more.Reference:
- Sarbacker GB, Wilder AG, Heart Failure Guidelines: Introduction to the New Agents. US Pharm. 2018; 43(2):22-26.