Pluripotent stem cells have been converted into functioning muscle tissue that can contract and respond to electrical and biochemical stimuli.
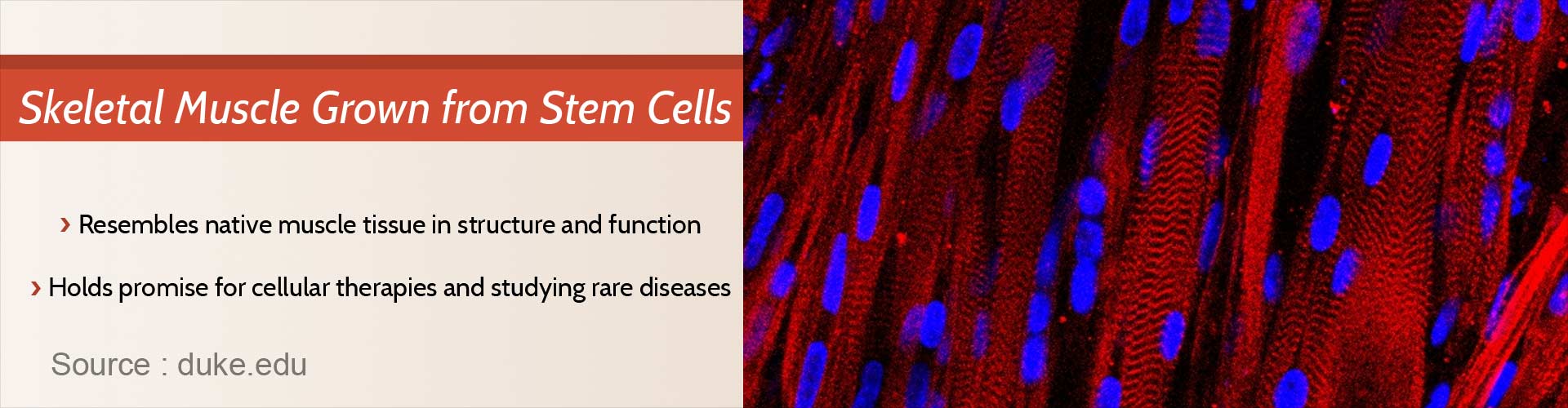
- Functioning human skeletal muscle has been produced from induced pluripotent stem cells taken from non-muscle cells of the skin and blood.
- The final muscle fiber is able to contract and respond to electrical and biochemical signals just like native tissue.
- The current method will prove useful in making healthy muscle in patients with rare muscle diseases.
TOP INSIGHT
Functioning human muscle has been grown for the first time from stem cells that can help cellular therapies, drug discovery and to study about rare diseases
Induced Pluripotent Stem Cells to Functioning Skeletal Muscles
- In the study, cells were taken from adult non-muscle tissues like skin and blood and reprogrammed to convert them back into naive or primitive cells
- These pluripotent stem cells were then grown in a medium that contained a molecule called Pax7 (a transcription factor), which regulates the proliferation of the muscle precursor cells to muscle cells
- After cell proliferation, the cells resembled adult muscle stem cells in structure. Now the goal was to convert these intermediate cells into functioning skeletal muscle
- In order to achieve this, the supply of the signaling molecule Pax7 was stopped and the cells were provided with a cell culture 3-D nutrient medium that gave them the support and nourishment to flourish into functioning muscle cells
- After two to four weeks of exposure to the 3-D culture, the muscle cells formed muscle fibers that were able to contract and respond to external stimuli such as electrical pulses and biochemical signals mimicking neuronal inputs just like native muscle tissue
- These newly grown muscle fibers, when introduced into adult mice, survived and functioned for at least three weeks while progressively integrating into the body’s native tissue through vascularization.
Making Functioning Muscle Fibers from Myoblasts
In Bursac and his team’s earlier research, which formed the basis of the current study, they had achieved to make functional muscle from small samples of human cells called "myoblasts” which they obtained from muscle biopsies; these cells are a step beyond the stem cell stage but are not yet mature muscle tissue. They grew the myoblasts several folds and then put them into a supportive 3-D scaffolding filled with a nourishing gel that allowed them to form aligned and functioning human muscle fibers.Advantages and Disadvantages of Muscle Tissue Grown from iPSCs
In the pathway taken to make muscle fibers from a pluripotent stem cell, reservoirs of "satellite-like cells" are developed, which help the normal adult muscles to repair damage, clearly proving to be an advantage over the method where muscle tissue is developed from myoblasts.Another advantage is that the stem cell method can also produce a greater quantity of muscle cells from a smaller starting batch compared to the biopsy method.
These advantages provide an easier path to genome editing and cellular therapies, and to develop tailored models of rare muscle diseases for drug discovery and basic biology studies and individualized health care.
Disadvantages of the stem cell method include the fact that the resulting muscle is less strong compared to native muscle tissue as well as the muscle grown from muscle biopsies.
However, on the whole, the current method can definitely be put to good use; one example is to use this technique along with genetic therapies - genetic malfunctions in the iPS cells derived from a patient can be fixed prior to growing them into mature muscle cells, thus producing completely healthy muscle.
"The prospect of studying rare diseases is especially exciting for us," said Bursac. "When a child's muscles are already withering away from something like Duchenne muscular dystrophy, it would not be ethical to take muscle samples from them and do further damage. But with this technique, we can just take a small sample of non-muscle tissue, like skin or blood, revert the obtained cells to a pluripotent state, and eventually grow an endless amount of functioning muscle fibers to test."
The research team is now fine-tuning their technique to grow more robust muscles and beginning work to develop new models of rare muscle diseases.
References:
- Lingjun Rao, Ying Qian, Alastair Khodabukus, Thomas Ribar and Nenad Bursac.Engineering Human Pluripotent Stem Cells Into a Functional Skeletal Muscle Tissue. Nature Communications, (2018), DOI: 10.1038/s41467-017-02636-4
- Lauran Madden, Mark Juhas, William E Kraus, George A Truskey, Nenad Bursac. Bioengineered human myobundles mimic clinical responses of skeletal muscle to drugs. eLife, (2015), DOI: 10.7554/eLife.04885
- Contracting human muscle tissue created - (http://serious-science.org/contracting-human-muscle-tissue-created-2203)
 MEDINDIA
MEDINDIA



 Email
Email










