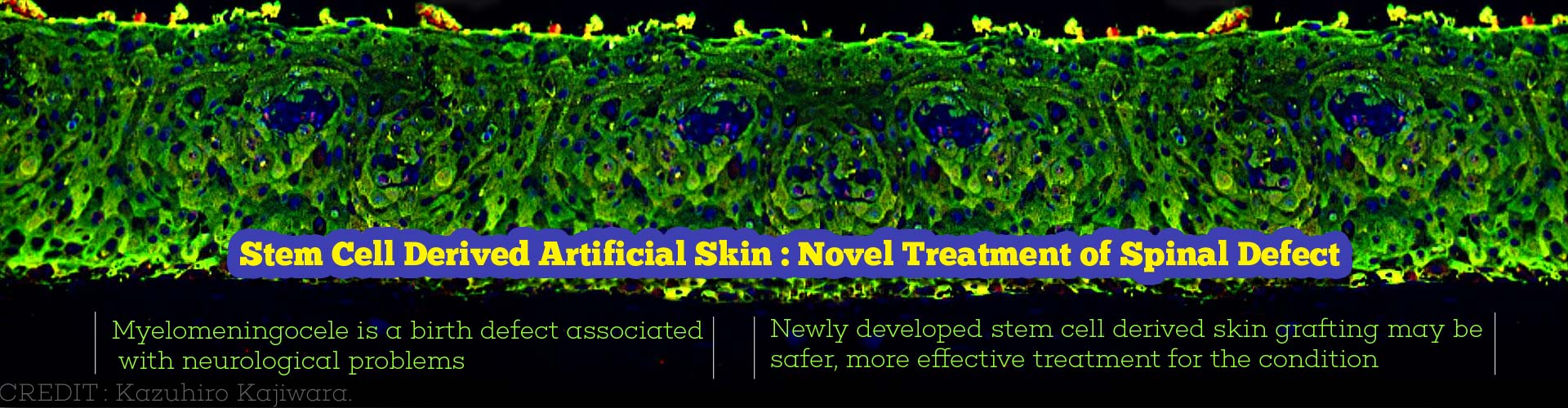
Artificial 3D skin produced from induced pluripotent stem cells successfully treats myelomeningocele, a spinal birth defect that causes neurological issues.
- Myelomeningocele is a birth defect of the spine associated with neurological problems.
- Current treatment options are associated with certain risks and limitations.
- Artificial skin generated from induced pluripotent stem cells (iPSCs) hopes to overcome these gaps.
The Necessity of New Stem Cell Therapies For Myelomeningocele
Usually, babies born with myelomeningocele undergo surgery to repair the defect within the first few days after birth, although the results may not be optimal. Some centers perform intrauterine surgery, which reportedly have a better outcome than post-natal surgery. However, intrauterine surgery is associated with an increased incidence of premature birth and other complications, making it necessary to explore safer and effective alternative prenatal treatments. To this end, Umezawa and his team set out to develop a minimally invasive technique to produce and transplant artificial skin grafts that could cover large defects earlier in the prenatal period, potentially aiming at better long-term prognosis while at the same time reducing surgical risks.TOP INSIGHT
Treatment of myelomeningocele prenatally at 28 weeks using artificial skin, appears a safer and more effective option than current therapies.
Generating 3D Skin using iPSCs To Treat Myelomeningocele
- Firstly the team obtained fetal cells from amniotic fluid from two pregnancies with severe fetal disease.
- These fetal cells were then genetically reprogrammed to become pluripotent stem cells, hence called induced pluripotent stem cells
- Using a chemical cocktail, by a novel protocol, these iPSCs were made to produce skin cells.
- The skin cells were further induced to proliferate and become multilayered 3D skin using growth factors such as epidermal growth factor.
- The entire process starting from amniotic fluid preparation to produce the artificial 3D skin took about 14 weeks.
- This would permit transplant of the skin into the fetus at 28-29 weeks of gestation.
Testing The 3D Artificial Skin On Rats
- The 3D skin grafts were transplanted into 20 rat fetuses via a small incision on the uterine wall.
- The 3D skin graft completely covered the myelomeningocele defects in 4, and partially covered them in 8 of the newborn rats, thereby protecting the spinal cord from direct exposure to harmful effects of chemicals and other harmful agents in the external environment.
- Interestingly, the engrafted skin regenerated with the growth of the fetus and provided skin coverage throughout the pregnancy period.
- Notably, the transplanted skin cells did not lead to tumor formation.
- However, the treatment significantly reduced birth weight and body length.
What Is Myelomeningocele?
Myelomeningocele, the most serious and common form of spina bifida, is a neural tube defect where the bones of the spine are completely formed. Consequently, portions of the spinal cord and nerves come out through the defect. Babies born with this disorder typically have an exposed area or a fluid-filled sac on the mid to lower back. Most children with this condition are at risk of brain damage since excess fluid builds up in their brains. They also suffer from symptoms such as loss of bladder or bowel control, numbness over the legs or feet, weakness and paralysis of the legs.What Are Pluripotent Stem Cells?
Induced pluripotent stem cells (iPSCs) are produced by genetically reprogramming skin or blood cells. These iPSCs are capable of differentiating into any type of human cell or tissue and have thus become an inexhaustible source of such cells for therapeutic purposes.The generation and use of iPSCs overcomes the ethical issues surrounding the use of embryonic stem cells for research and treatment purposes.
The use of iPSCs was pioneered by Shinya Yamanaka from Japan, for which he was awarded the Nobel Prize in 2012.
The current study illustrates once again the importance of stem cells and tissue engineering technology in treating several diseases.
Conclusion
Dr Umezawa says, "We are encouraged by our results and believe that our fetal stem cell therapy has great potential to become a novel treatment for myelomeningocele. However, additional studies in larger animals are needed to demonstrate that our fetal stem cell therapy safely promotes long-term skin regeneration and neurological improvement."References:
- Kazuhiro Kajiwara, Tomohiro Tanemoto, Seiji Wada, Jurii Karibe, Norimasa Ihara, Yu Ikemoto, Tomoyuki Kawasaki, Yoshie Oishi, Osamu Samura, Kohji Okamura, Shuji Takada, Hidenori Akutsu, Haruhiko Sago, Aikou Okamoto, Akihiro Umezawa. Fetal Therapy Model of Myelomeningocele with Three-Dimensional Skin Using Amniotic Fluid Cell-Derived Induced Pluripotent Stem Cells. Stem Cell Reports (2017) DOI: http://dx.doi.org/10.1016/j.stemcr.2017.05.0
 MEDINDIA
MEDINDIA




 Email
Email