Deferoxamine, used to treat iron overload, can be used to prevent leukemia cells from overtaking stem cell sites in the bone marrow.
- Adding deferoxamine to chemotherapy treatment for Acute Myeloid Leukaemia (AML) increases survival prospects.
- Deferoxamine, a drug used to treat iron overload, blocks leukemia cells from invading stem cell areas in the bone marrow.
- The drug facilitated the rescue of healthy blood stem cells thus improving the efficiency of chemotherapy.
Study Overview
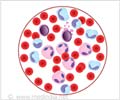
The research team has visualized how leukemia cells infiltrate the bone marrow in mice and have made a crucial discovery. There are certain specific areas or blood vessels of the bone marrow that support stem cells that differentiate into all types of blood cells. However, it was observed that when the leukemia cells take over these stem cell areas, the stem cell population is lost and so is the production of healthy blood cells. ‘Hijacking’ these stem cell sites causes problems like anemia, infection and bleeding in patients that affects the success of chemotherapy.Another important finding was that a drug already approved to treat a condition called iron overload can protect stem cell areas in the bone marrow from being overtaken by the cancer cells.
"Since the drug is already approved for human use for a different condition, we already know that it is safe. We still need to test it in the context of leukemia and chemotherapy, but because it is already in use we can progress to clinical trials much quicker than we could with a brand new drug," said lead author, Dr Cristina Lo Celso from the Department of Life Sciences at Imperial.
Human Clinical Trials
Since the drug is already an FDA approved drug, the time required to bring it into the market if found appropriate for AML is significantly less. Understanding if deferoxamine is an option for treating AML in humans should take less than 5 years, compared to 10 to 15 years for an entirely new drug.Deferoxamine has also been used to treat myelodysplasia, a leukemia-related disease where young blood stem cells do not mature into healthy blood cells.
The research team aims to collaborate with clinicians and begin human clinical trials of the drug for treating AML.
References:
- Delfim Duarte et al. Inhibition of endosteal vascular niche remodeling rescues hematopoietic stem cell loss in AML. Cell Stem Cell. https://doi.org/10.1016/j.stem.2017.11.006