Zurampic (lesinurad) is a drug to treat high levels of uric acid in the blood (hyperuricemia) associated with gout disease.

‘Zurampic gained FDA approval in combination with a xanthine oxidase inhibitor (XOI) for the treatment of hyperuricemia (high levels of uric acid in the blood) associated with gout.’





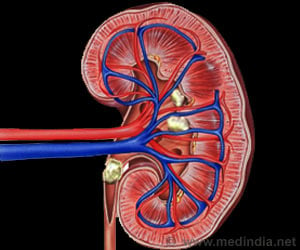
"Controlling hyperuricemia is critical to the long-term treatment of gout. Zurampic provides a new treatment option for the millions of people who may develop gout over their lifetimes,” said Dr.Badrul Chowdhury, director of the Division of Pulmonary, Allergy and Rheumatology Products in the FDA’s Center for Drug Evaluation and Research.Zurampic works by helping the kidney excrete uric acid. It does this by inhibiting the function of transporter proteins involved in uric acid reabsorption in the kidney.
FDA has approved the drug with a box warning as it has a risk of acute kidney (renal) failure, which is more common when used without an XOI and with higher than approved doses of Zurampic.
Source-Medindia