For patients with relapsing-remitting multiple sclerosis, high-dose immunosuppressive therapy with autologous hematopoietic cell transplant is related with remission and improvement.
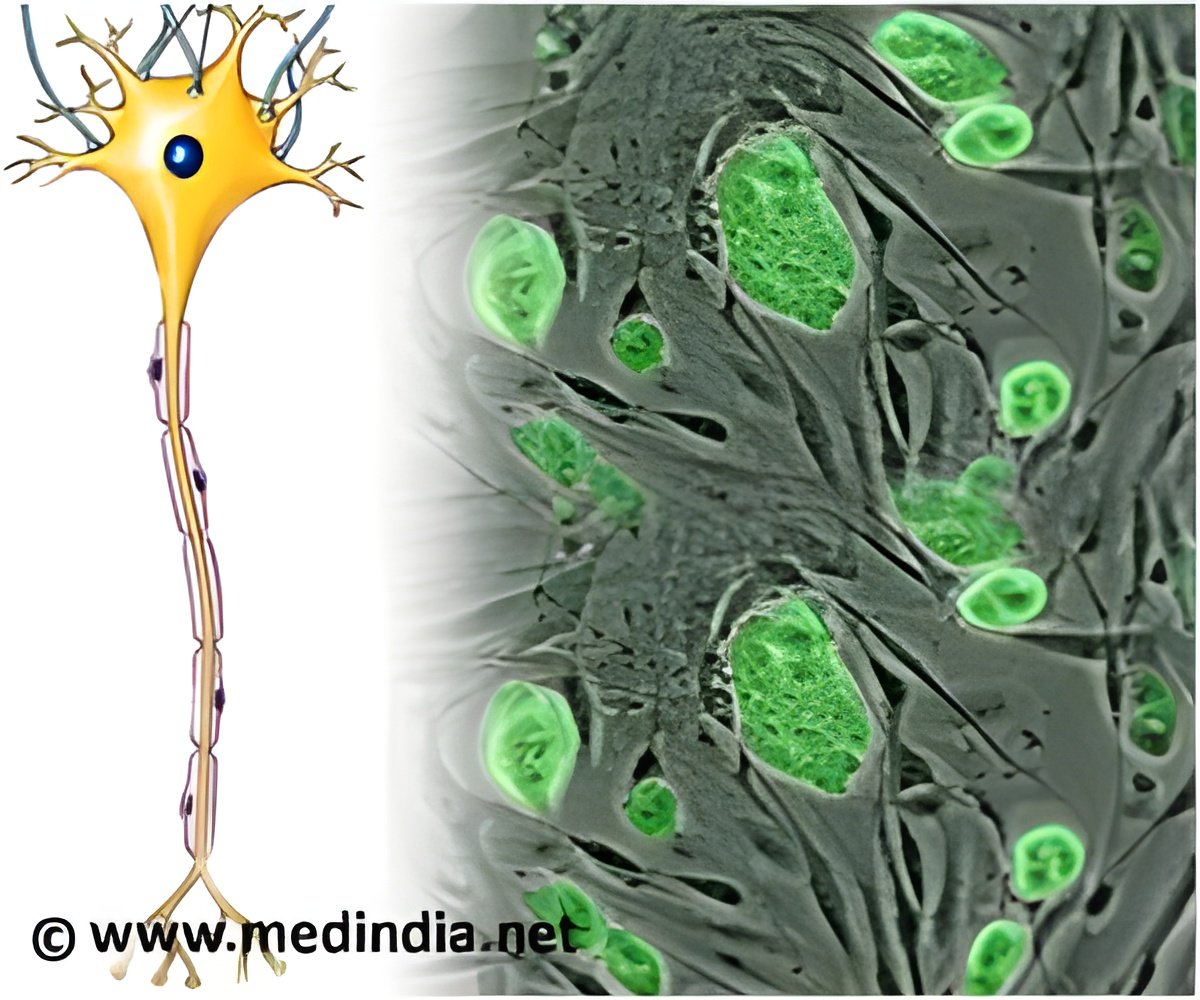
Researchers examined the effectiveness of early intervention with HDIT/HCT for patients with RRMS and breakthrough disease. They found that of the 24 patients who received HDIT/HCT, the overall rate of event-free survival was 78.4 percent at 3-years, which was defined as survival without death or disease from a loss of neurologic function, clinical relapse or new lesions observed on imaging, while progression-free survival and clinical relapse-free survival were 90.9 percent and 86.3 percent, respectively, at 3-years. The adverse events were consistent with the expected toxic effect of HDIT/HCT and no acute treatment-related neurologic adverse events were seen. Researchers also noted improvements in neurologic disability, quality-of-life and functional scores.
The study is titled 'The Hematopoietic Cell Transplantation for Relapsing-Remitting Multiple Sclerosis (HALT-MS)'.
Source-Medindia
 MEDINDIA
MEDINDIA

 Email
Email









