Genetic predisposition to the progression of liver damage through the AEBP1 gene affects the development, severity, and potential treatment of liver disease, says TGen-led study.

‘Expression of AEBP1 gene is linked to the onset and severity of fibrosis and may be a therapeutic target for the progression of liver damage.’
Read More..




The findings are detailed in a study published in the scientific journal PLoS One.Read More..
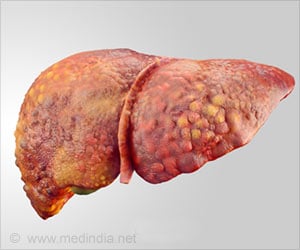
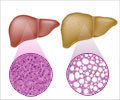
The study results suggest that increased expression of AEBP1 correlates with the severity of liver fibrosis in patients with NASH (nonalcoholic steatohepatitis), which is a type of NAFLD (nonalcoholic fatty liver disease), the most common cause of liver damage. NASH indicates there is both inflammation and liver cell damage, along with fat in the liver.
"Given the strong link between fibrosis and risk of liver-related mortality, efforts to identify and characterize the specific mechanisms contributing to NAFLD progression are critical for the development of effective therapeutic and preventative strategies," said Dr. Johanna DiStefano, head of the Diabetes and Fibrotic Disease Unit at TGen.
One of the study's major findings is that AEBP1 regulates the expression of a network of at least nine genes related to fibrosis: AKR1B10, CCDC80, DPT, EFEMP1, ITGBL1, LAMC3, MOXD1, SPP1, and STMN2.
"These findings indicate that AEBP1 may be a central regulator of a complex fibrosis gene expression network in the human liver," said Dr. DiStefano, the study's senior author.
Advertisement
- AEBP1 contributes to liver fibrosis by modulating a gene network specific to stellate cells, the key fibrogenic cells of the liver.
- AEBP1 expression is increased by obesity-related factors linked to NAFLD, including the sugars glucose and fructose, and palmitate, a fatty acid commonly found in processed foods.
- Two microRNAs -- miR-372-3p and miR-373-3p -- which otherwise limit the expression of AEBP1, are reduced in NASH patients with advanced fibrosis. MicroRNAs constitute a recently discovered class of non-coding RNAs that play key roles in the regulation of gene expression.
"Our work shows that AEBP1 parallels the onset and severity of fibrosis in NASH patients, and suggest that AEBP1 may represent a specific therapeutic target to prevent the development of NASH fibrosis," said Dr. Glenn S. Gerhard, Chair of the Department of Medical Genetics and Molecular Biochemistry at the Lewis Katz School of Medicine at Temple University. Dr. Gerhard is the study's lead author.
Advertisement
Source-Eurekalert