As a gene responsible for Thanatophoric Dysplasia, fibroblast growth factor receptor 3 (FGFR3) has been reported.
TOP INSIGHT
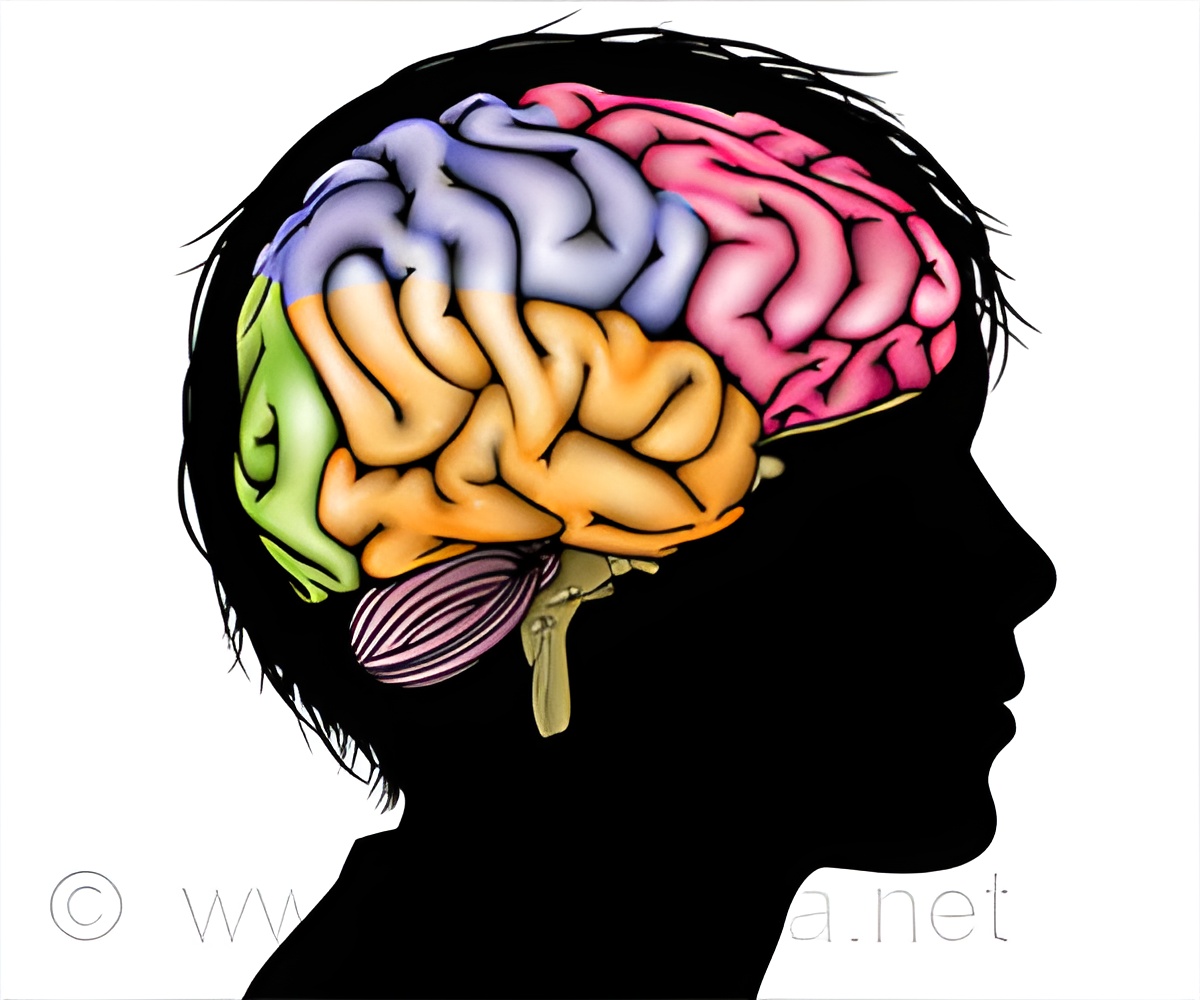
Since Thanatophoric dysplasia (TD) is a rare disease and since it is difficult to obtain brain samples from human patients, the pathophysiology of TD is largely unknown, and effective therapy has not been established.
The present research team of Kanazawa University succeeded in generating an animal model using ferrets, which reproduces the brain abnormalities found in human TD patients. By using this animal model, the team elucidated the formation process of polymicrogyria, one of the abnormalities found in the TD brain. In the present study, the team has investigated the formation process of PNH, the other brain abnormality found in human TD patients, using their animal model of TD.
The present research team has succeeded in uncovering the pathophysiological process of the formation of PNH by using the TD ferrets as follows.
First, PNH was analyzed in terms of composing cell types to reveal that a large number of neurons but few glial cells existed in PNH. In healthy brain, neurons are found in the cerebral cortex near the brain surface. It was, therefore, considered that during the fetal brain development, PNH formation might be induced by inability of neurons to translocate themselves to their destination, that is, the cerebral cortex. Next, the reason why neurons were unable to translocate themselves was investigated. It was found that spatial arrangement of radial glial cells were distorted; radial glial fibers were considered to serve as the track for neurons to translocate themselves. Thus, it was suggested that the distortion of radial glial fibers would be a reason for aberrant localization of neurons.
The research team elucidated the important process of the PNH formation mechanism. It is only possible, by using appropriate animal model that reproduces relevant pathophysiology, to uncover the process of pathogenesis and to develop therapy. Since the research on abnormalities of bones in TD is progressing with iPS cells at Kyoto University, it is expected that the whole aspect of TD with brain and bone abnormalities would be elucidated and that the therapeutic methods would be developed.
Source-Eurekalert
 MEDINDIA
MEDINDIA



 Email
Email









