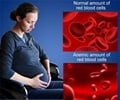
An important DNA repair protein, p53, mediates many facets of Diamond Blackfan Anemia. But, its mechanism has not, until now, been well understood.

TOP INSIGHT
A deficiency in RPS19, the most commonly mutated ribosomal protein in Diamond Blackfan Anemia (DBA) patients, leads to the upregulation and activation of the p53 pathway.
In a recently published article in Disease Models & Mechanisms, Dr. Sakamoto's team used a zebrafish model of DBA with RPS19 and RPL11 insufficiency to further characterize the link between defects in ribosome biogenesis, nucleotide metabolism, and the p53 pathway in DBA.
The RPS19-deficient zebrafish showed a decrease in proliferation, enhanced activation of the ATR/ATM-CHK1/CHK2/p53 DNA damage pathway, an imbalanced pool of nucleotides, ATP depletion, and AMPK activation. These findings are all hallmarks of cellular energy crisis, DNA replication stress, and thus enhanced DNA repair. When treating zebrafish with exogenous nucleosides, a decrease in the activation of p53 and AMPK was observed.
As blood cells are highly dependent on salvage pathways for the production of nucleotides and are therefore vulnerable to a stressed metabolism, red blood cells in DBA patients may benefit from exogenous nucleosides. Nucleoside supplements are known to be very safe and are even included in many infant formulas. This form of supplementation may be beneficial, not only in patients with DBA, but also for other conditions that involve the activation of the DNA damage response, such as radiation exposure.
Furthermore, treatment of the RP-deficient zebrafish with inhibitors of various cell cycle checkpoint kinases decreased p53 upregulation and apoptosis while resulting in an improvement of hematopoiesis. Therefore drugs that work to decrease DNA damage or help increase DNA repair could be effective for the treatment of DBA.
Source-Eurekalert
 MEDINDIA
MEDINDIA

 Email
Email