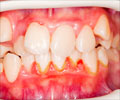
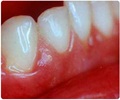
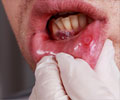
A gene that is common in the development of gum disease, rheumatoid arthritis, and osteoporosis has been identified by an international team of researchers.
Experts at Hospital for Special Surgery say that their findings about the gene, called interferon regulator factor-8 (IRF-8), may lead to new treatments in future."The study doesn't have immediate therapeutic applications, but it does open a new avenue of research that could help identify novel therapeutic approaches or interventions to treat diseases such as periodontitis, rheumatoid arthritis or osteoporosis," said Nature magazine quoted Dr. Baohong Zhao, a research fellow in the Arthritis and Tissue Degeneration Program at Hospital for Special Surgery located in New York City, as saying.
The researchers discovered that downregulation of IRF-8 (meaning that the gene produces less IRF-8 protein) increases the production of cells called osteoclasts that are responsible for breaking down bone.
In humans and animals, bone formation and bone resorption are closely coupled processes involved in the normal remodelling of bone. Enhanced development of osteoclasts, however, can create canals and cavities that are hallmarks of diseases such as periodontitis, osteoporosis and rheumatoid arthritis.
The genome-wide study showed that the expression of IRF-8 was reduced by 75 percent in the initial phases of osteoclast development.
The genetically engineered mice deficient in IRF-8 had decreased bone mass and severe osteoporosis.
Advertisement
"This is the first paper to identify that IRF-8 is a novel key inhibitory factor in osteoclastogenesis (production of osteoclasts)," said Zhao.
Advertisement
The study has been published in the journal Nature Medicine.
Source-ANI
RAS