"A bioartificial ovary has the potential to secrete hormones in a natural way based on the body's needs, rather than the patient taking a specific dose of drugs each day," Emmanuel C. Opara, Ph.D., professor of regenerative medicine Wake Forest Baptist and senior researcher, said.
Tamer Yalcinkaya, M.D., associate professor and section head of reproductive medicine at Wake Forest Baptist, said "This research project is interesting because it offers hope to replace natural ovarian hormones in women with premature ovarian failure or in women going through menopause."
"The graft format would bring certain advantages: it would eliminate pharmacokinetic variations of hormones when administered as drugs and would also allow body's feedback mechanisms to control the release of ovarian hormones," he said.
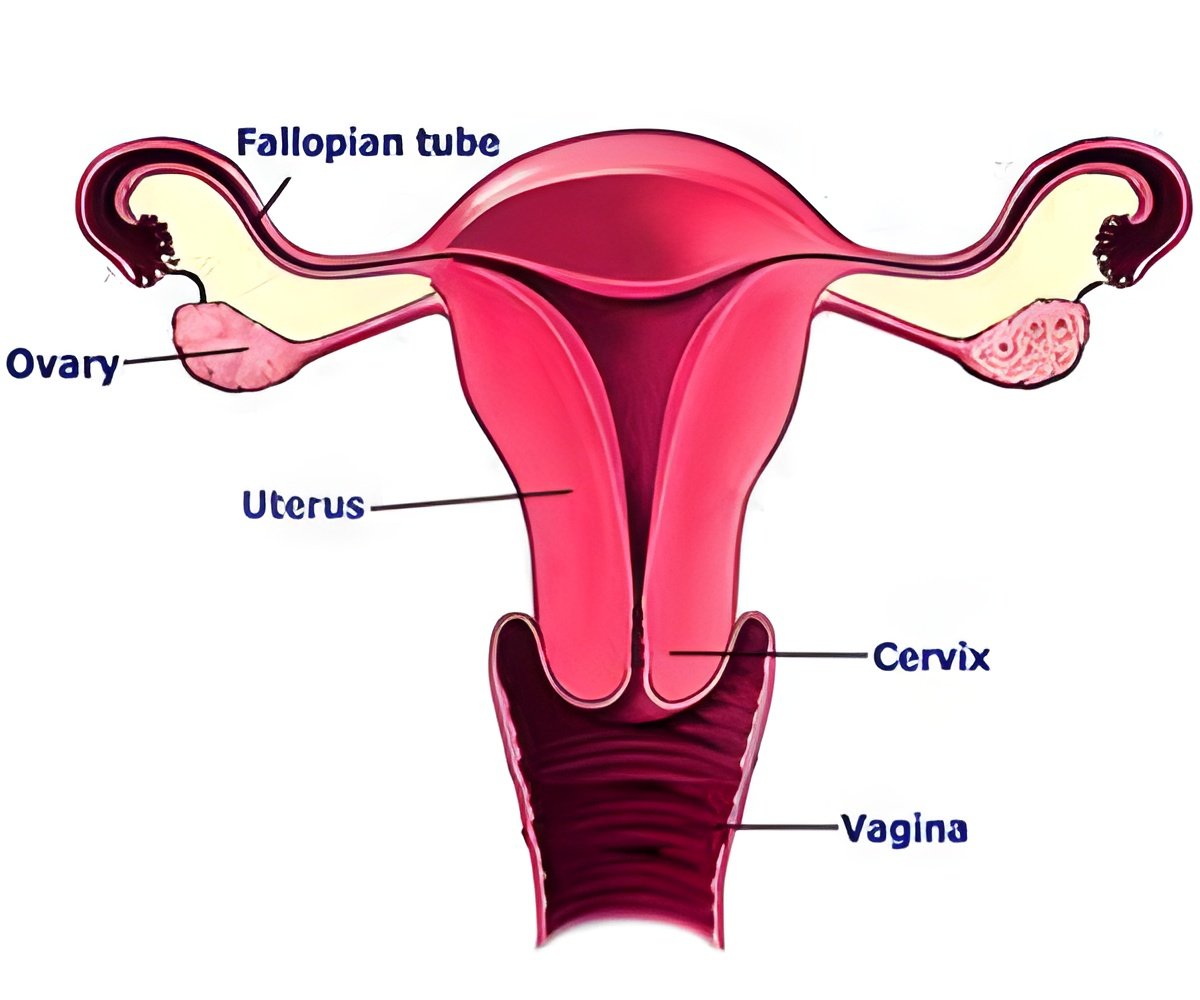
It involves encapsulating ovarian cells inside a thin membrane that allows oxygen and nutrients to enter the capsule, but prevents patients from rejecting the cells, with this scenario, functional ovarian tissue from donors can be used to engineer bioartificial ovaries with non-functioning ovaries.
The team isolated the two types of endocrine cells found in ovaries (theca and granulosa) from 21-day-old rats and the cells were encapsulated inside materials compatible with body.
Advertisement
The arrangement of cells that most closely mimicked the natural ovary (layers of cells in a 3-D shape) secreted levels of estrogen that were 10 times higher than other cell arrangements.
Advertisement
The research has been published in Biomaterials.
Source-ANI