"Of the nine patients observed in the study, three patients were able to read letters spontaneously," Retina Implant AG, a nine-year-old technology startup company that invented the device, said in a press release.
"During observation in and outside the laboratory, patients also reported the ability to recognise faces, distinguish objects such as telephones and read signs on doors."
The study appears in Proceedings of the Royal Society B, a journal published by Britain's de-facto academy of sciences.
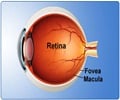
The device consists of a tiny light-sensitive chip measuring 3mm by 3mm (0.11 x 0.11 inches), which sends electrical signals down the optic nerve to the brain, providing a "diamond-shaped" black-and-white image with a field of 15 degrees.
Attached to the retina, the implant is powered via a thin cable which connects to a small coil fitted under a fold of skin behind the ear.
Advertisement
The battery unit also has two knobs, enabling the user to adjust the brightness and contrast of the image.
Advertisement
One of the nine had to drop out of the experiment after the optic nerve was damaged during the implant operation, and another experienced a buildup in eyeball pressure which was successfully treated with drugs.
New drugs and revolutionary medical devices typically undergo a three-phase process of trials on human volunteers.
The number of patients and the scope of the test gradually widens, in a bid to ensure that the innovation is both safe and effective.
The first trial of the implant, published in 2010, used a cable, rather than wireless technology, to power the device.
There are several other entrants in the field for retinal implants, reflecting big advances in electronic miniaturisation and microsurgery in the past decade.
None claims to be a cure but rather an aid to distinguish between light and darkness and ascertain the shape of objects.
"Although the restoration of vision described here is limited, blind persons with no alternative therapy options regard this type of artificial vision as an improvement in everyday life," the German doctors said.
Last week the US firm Second Sight Medical Products gained US regulatory approval in addition to the green light from Europe for its Argus II retinal prosthesis.
There is also a 24-electrode device made by Bionic Vision Australia, which has so far been tested on one patient.
Source-AFP