Enzyme USP15 may have role in future treatment of breast and pancreatic cancers, said researchers.
TOP INSIGHT
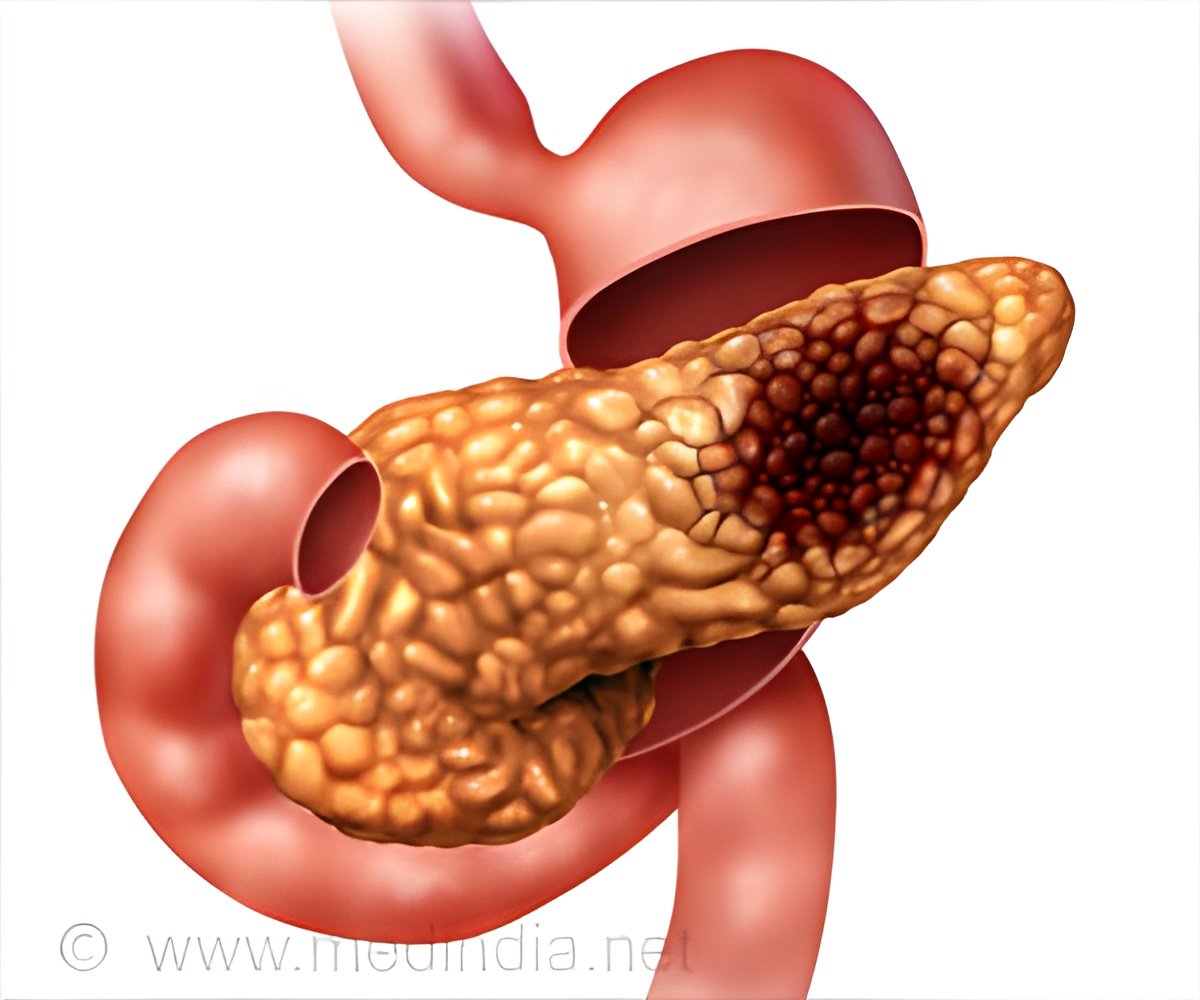
The Cancer Genome Atlas indicates that USP15 enzyme deletions occur in 16 percent of breast cancers and in 5 percent of pancreatic cancers.
PARP inhibitors are a new class of pharmacological inhibitors developed for multiple purposes, but chiefly for the treatment of cancer. They have garnered a great deal of attention for their potential in the management of patients with BRCA mutations.
Pei and his research team found that USP15 regulates homologous recombination -- one of the major pathways to repair DNA damage affecting broth strands of the double helix -- and cancer cell response to PARP inhibitors.
USP15 is part of a group of deubiquitinating enzymes, responsible for removing ubiquitin chains from proteins and other molecules, which play important roles in maintaining genome stability. Based on their research, Pei and his team believe USP15 may function similarly to the USP4 enzyme, which was found to play a role in DNA repair by Pei's group four years ago.
"USP15 is a potential biomarker for treatments of pancreatic cancer, as well as breast and ovarian cancers," explained Yihan Peng, a PhD student in Pei's lab and the first author on the study.
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email










