There is need of an alternative approach to endodontic treatment, with the goal of regenerating a damaged tooth so that it functions like any other normal tooth.
TOP INSIGHT
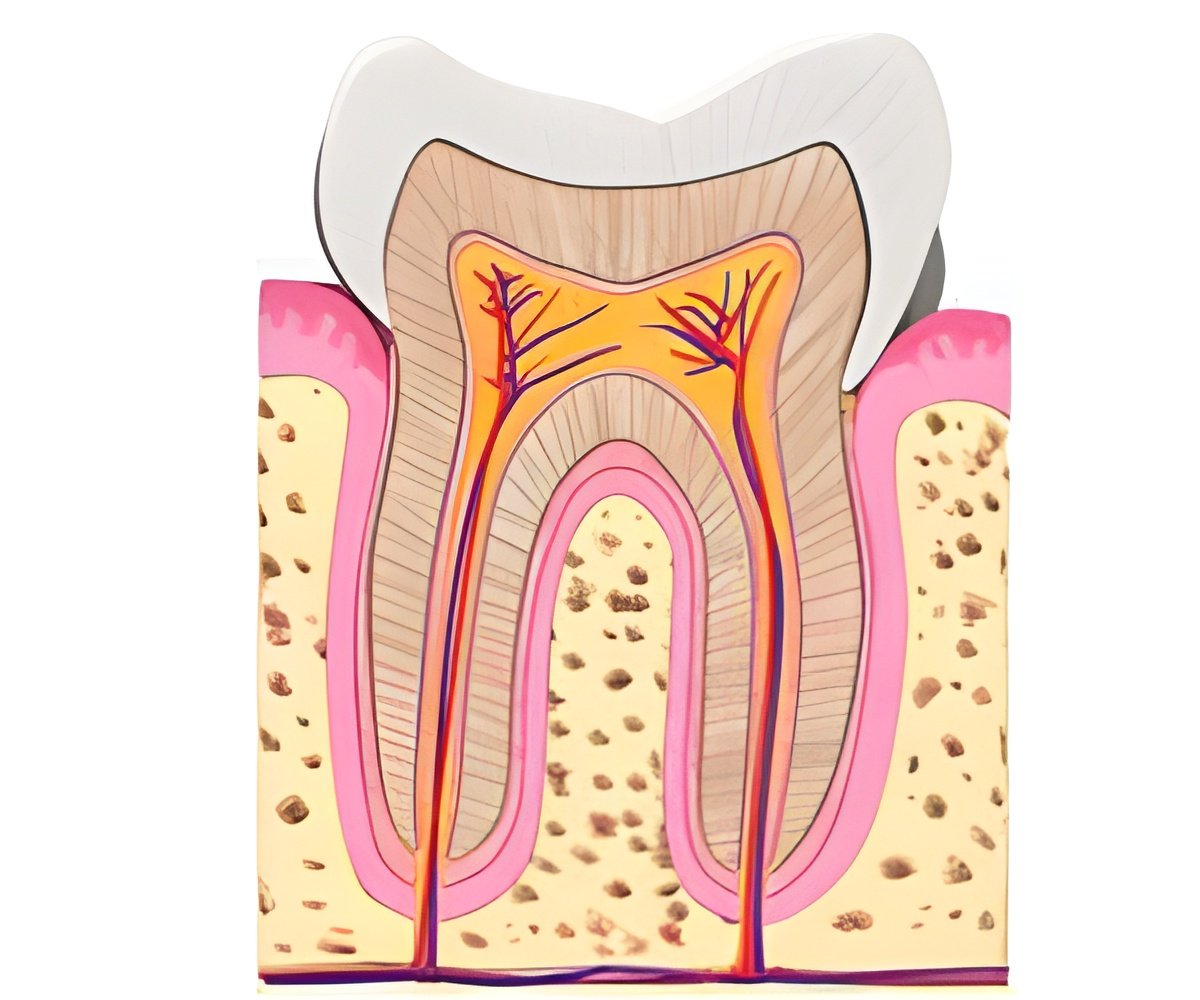
Using a collagen-based biomaterial to deliver stem cells inside damaged teeth can regenerate dental pulp-like tissues in animal model experiments.
Yelick and her colleagues, including lead study author Arwa Khayat, former graduate student in dental research at TUSDM, examined the safety and efficacy of gelatin methacrylate (GelMA) - a low-cost hydrogel derived from naturally occurring collagen - as a scaffold to support the growth of new dental pulp tissue.
Using GelMA, the team encapsulated a mix of human dental pulp stem cells - obtained from extracted wisdom teeth - and endothelial cells, which accelerate cell growth. This mix was delivered into isolated, previously damaged human tooth roots, which were extracted from patients as part of unrelated clinical treatment and sterilized of remaining living tissue. The roots were then implanted and allowed to grow in a rodent animal model for up to eight weeks.
The researchers observed pulp-like tissue inside the once empty tooth roots after two weeks. Increased cell growth and the formation of blood vessels occurred after four weeks. At the eight-week mark, pulp-like tissue filled the entire dental pulp space, complete with highly organized blood vessels populated with red blood cells.
The team also observed the formation of cellular extensions and strong adhesion into dentin - the hard, bony tissue that forms the bulk of a tooth. The team saw no inflammation at the site of implantation, and found no inflammatory cells inside implanted tooth roots, which verified the biocompatibility of GelMA.
The results support GelMA-encapsulated human dental stem cells and umbilical vein endothelial cells as part of a promising strategy to restore normal tooth function, according to the study authors. However, they note that the current study was limited to partial tooth roots and did not examine nerve formation in regenerated dental pulp tissue. They emphasize the need for additional safety and efficacy studies in larger animal models before human clinical trials can be considered.
Source-Eurekalert
 MEDINDIA
MEDINDIA


 Email
Email