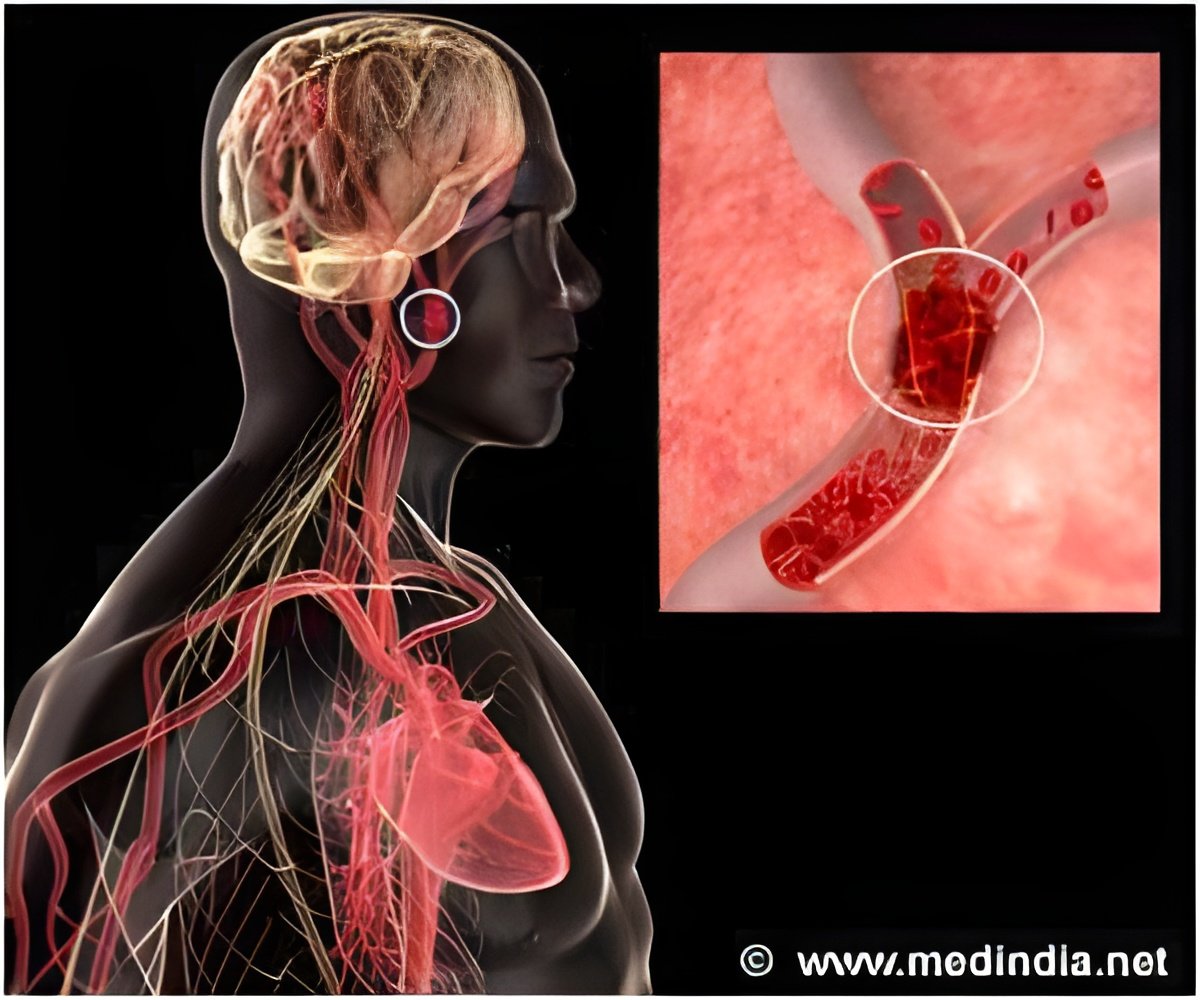
It has a self-expanding, stent-like design, and once inserted into a blocked artery using a thin catheter tube, it compresses and traps the clot. The clot is then removed by withdrawing the device, reopening the blocked blood vessel, the British journal The Lancet reports.
"This new device is significantly changing the way we can treat ischemic stroke (caused by blockage of an artery to the brain)," said Jeffrey L. Saver, director of the University of California Los Angeles Stroke Centre and a professor of neurology at the David Geffen School of Medicine.
"We are going from our first generation of clot-removing procedures, which were only moderately good in reopening target arteries, to now having a highly effective tool," added Saver, who led the study, according to a California statement.
Results of the study showed that the device opened blocked vessels without causing symptomatic bleeding in or around the brain in 61 percent of patients.
The standard FDA-approved mechanical device, a corkscrew-type clot remover called the MERCI Retriever, was effective in 24 percent of cases. The use of SOLITAIRE also led to better survival three months after a stroke.
Advertisement
About 87 percent of all strokes are caused by blood clots blocking a blood vessel supplying the brain.
Advertisement
When clot-busting drugs cannot be used or are ineffective, the clot can sometimes be mechanically removed during, or beyond, the four-and-a-half-hour window.
The current study, however, did not compare mechanical clot removal to drug treatment.
Source-IANS