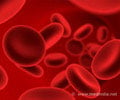
Human red blood cells have been developed for the first time from embryonic stem cells by Brtish scientists working on a project to manufacture synthetic blood on an industrial scale.
The project, funded by the Wellcome Trust, has used more than a hundred spare IVF embryos from fertility clinics to establish several embryonic stem cell lines that replicate continuously in the laboratory.
Professor Marc Turner, leader of the project, said that synthetic blood made on an industrial scale would overcome the problem of blood shortages and the risk of transmitting new infections between donors and recipients.
"We've proved the principle that from these embryonic stem cell lines we can generate red blood cells," the Independent quoted Turner as saying.
"At the end of this three-year period we would envisage generating up to a unit [a pint] of red cells from embryonic stem cells at clinical grade which fulfil all the in vitro characteristics of red cells."
As part of the process of gaining regulatory approval, the project has produced four embryonic stem cell lines deemed good enough to be used in human clinical trials - conforming to the "good manufacturing practice" (GMP) grade.
Advertisement
"We have cells that are clearly red so we're happy with that. We've managed to go 90 per cent down the path towards fully differentiated, adult red blood cells," said Mountford.
Advertisement
"We're confident that we'll get the numbers of mature red blood cells that we need for clinical trials," she said.
Only one of the four embryonic stem cells lines produced at GMP grade has been tested for its blood type, Professor Turner said.
The line, known as RC-9, is blood type B-positive and although it is not the universal donor type it can still be used for research.
Source-ANI