When 334 people were treated with the device it showed that 99% had no difficulty with clarity of vision and 97% had no difficulty with their daily activities.

"The iDesign System for LASIK is a leap forward for laser vision correction in the United States, enabling a highly personalized treatment unique to the vision needs of each person," said Leonard Borrmann, head of research and development for Abbott's vision business.
In a study where 334 eyes were treated with the iDesign System found that, 99% of patients had little or no difficulty with the clarity of their vision and 97% of patients had little or no difficulty with their daily activities after surgery. Also, remarkable improvements in vision while driving at night and a reduction in glare was noticed by these patients.
"The iDesign System allows doctors to detect details of the eye that we were never able to see before," said Edward E. Manche, director of Cornea and Refractive Surgery and professor of Ophthalmology at Stanford University School of Medicine. “After surgery, people who were treated with the iDesign System in a clinical study reported high satisfaction rates, as well as improvements in all areas of vision well-being, such as their quality of vision and vision needed for healthy, active lifestyles.”
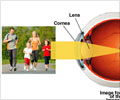
More people may now become eligible for LASIK. This system will aid doctors in treating people who have higher levels of astigmatism with nearsightedness and a range of pupil sizes.
James Webb Space Telescope will be integrated with the imaging technology developed at Abbott and put in orbit in 2018.
 MEDINDIA
MEDINDIA



 Email
Email