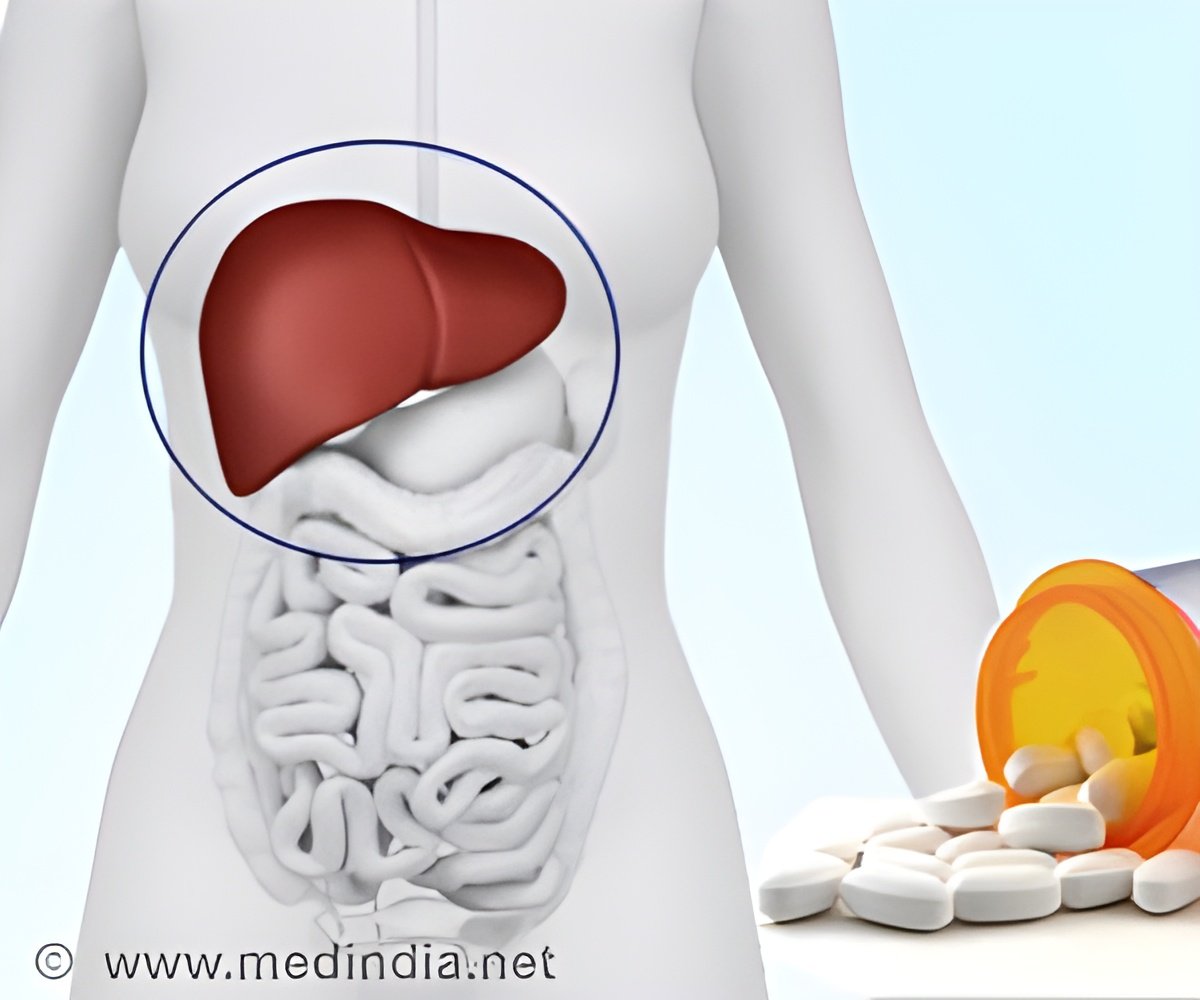
At least 26 cases of liver damage that may be tied to hepatitis C drug Viekira Pak or Technivie have been reported worldwide since the drugs were approved.
Hepatitis C is an infectious disease usually spread by unsanitary injection practices. The disease targets the liver, though it is only rarely life-threatening. Viekira Pak or Technivie are often used to treat the infection. However, the US Food and Drug Administration has warned that these two drugs manufactured by Chicago-based AbbVie could cause liver damage, leading in some cases to death.
Some patients using the either of the two drugs were found to have experienced liver failure or related conditions, including some that resulted in a liver transplant or death. The worst outcomes were observed mostly in patients who had pre-existing liver cirrhosis. The FDA said, "At least 26 cases of liver damage that may be tied to the two drugs have been reported worldwide since the drugs were approved."
The company said, "We are updating the safety risks on the drugs’ labels to emphasize that the drugs should not be used by those with advanced cirrhosis."
The FDA said, "Because post-marketing events are reported voluntarily during clinical practice, estimates of frequency cannot be made and a causal relationship between treatment and these events has not been established. The drugs can cause serious liver injury mostly in patients with underlying advanced liver disease. Patients should not stop taking the drugs without consulting their doctor since that could result in their hepatitis becoming resistant to other treatments."
Source-AFP
 MEDINDIA
MEDINDIA

 Email
Email