A new model system that can be used to develop drug therapies for genetic disorders like spinocerebellar ataxia type 6 , has been created.
- Spinocerebellar ataxia type 6 (SCA6) is a movement disorder characterized by death of Purkinje cells in the cerebellum.
- The disease results from mutations that increase the number of times a particular section of the CACNA1A gene are repeated.
- Researchers made a disease model based on human Purkinje cells grown in culture.
- Researchers discovered that mature Purkinje cells with the SCA6 mutation became vulnerable when deprived of thyroid hormone.
- The patient-derived Purkinje cells show a vulnerability to nutrient depletion and that this vulnerability can be suppressed by several compounds.
TOP INSIGHT
The patient-derived Purkinje cells grown in culture will be extremely useful for investigating disease mechanisms and for developing effective drug therapies.
The disease manifests in middle age and results from mutations that increase the number of times a particular section of the CACNA1A gene are repeated.
As an alternative, the team led by Keiko Muguruma focused their efforts on making a disease model based on human Purkinje cells grown in culture.
Published in Cell Reports, the study shows how stem cells from patients with SCA6 can be transformed into mature Purkinje cells, the same type of neuron that starts dying when people develop SCA6 later in life. With this setup, the team discovered that mature Purkinje cells with the SCA6 mutation became vulnerable when deprived of thyroid hormone.
As Muguruma explains, "we succeeded in generating Purkinje cells with full sets of SCA6 patient genes. Unlike animal models, these patient-derived Purkinje cells will be extremely useful for investigating disease mechanisms and for developing effective drug therapies."
When tested, the team found that while both types of mature Purkinje cells seemed outwardly similar, they differed in how much the CACNA1A gene was expressed.
The part of the CACNA1A protein that contains the excessively repeated section is called α1ACT.
When researchers compared expression of this fragment between normal and patient-derived cells, they found that it was expressed much less in the SCA6 Purkinje cells.
Because α1ACT normally binds to DNA in the nucleus and triggers the expression of other proteins that are important for normal Purkinje-cell development, these proteins were also expressed much less in the cells that contained the mutation.
Again, when the team looked at immature Purkinje cells, α1ACT expression was similar for all groups.
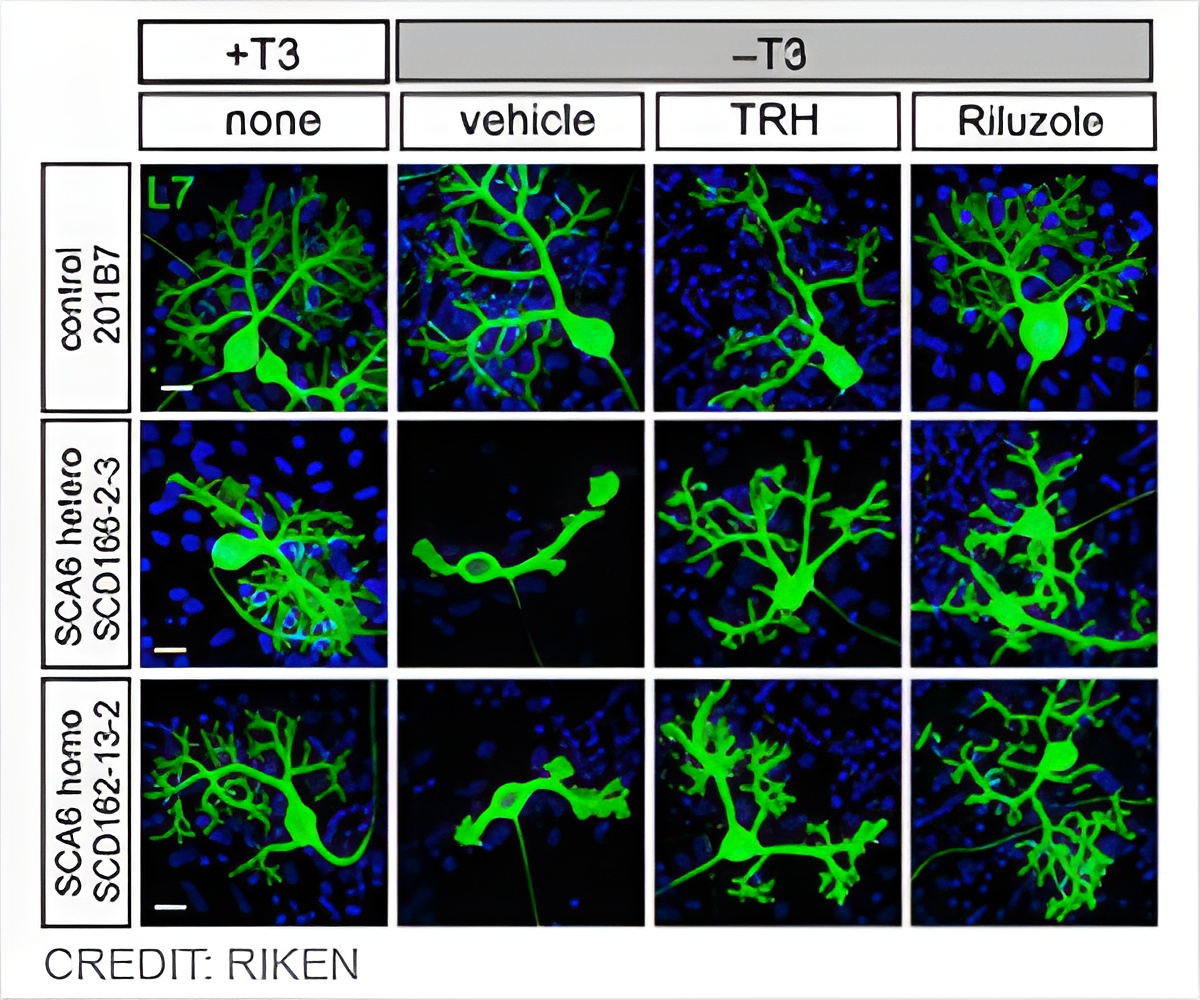
"This new system is particularly useful for drug discovery," notes Muguruma. "Using it, we were able to demonstrate that patient-derived Purkinje cells show a vulnerability to nutrient depletion and that this vulnerability can be suppressed by several compounds."
Knowing that thyroid hormone is important for proper maturation and maintenance of Purkinje cells, the researchers deprived mature neurons of the hormone and found that many of the patient-derived cells died, while those that survived showed physical abnormalities. Purkinje cells without the mutation were unaffected.
Muguruma cautions, "there are some reports that hypothyroidism is related to cerebellar ataxia and cerebellar atrophy, but we do not yet know whether the SCA6 disease phenotypes are causally linked to decreased thyroid hormone."
Now that they have proved the usefulness of this model system, Muguruma and her colleagues can continue to investigate how thyroid releasing hormone was able to protect the cells, and ultimately find a cure for this type of spinocerebellar ataxia.
Source-Medindia
 MEDINDIA
MEDINDIA



 Email
Email